
Concept explainers
(a)
Interpretation:
The reactions is to be written for the reaction of
Concept Introduction:
Hydration:
When
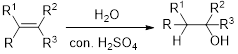
Alkene is reaction with water in the presence of sulfuric acid, first step is proton (
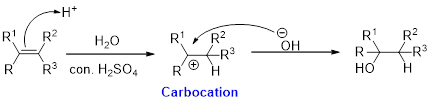
In hydration reaction, sulfuric acid is act as a proton donor, which is the driving force of the reaction. Hydration reaction will not go without acid (sulfuric acid).
(b)
Interpretation:
The reactions is to be written for the reaction of
Concept Introduction:
Hydrobromination:
A hydrobromination reaction is one of the electrophilic additions to alkenes to yield the corresponding bromo
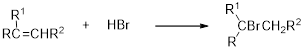
(b)
Interpretation:
The reaction is to be written for the reaction of
Concept Introduction:
An alkene undergo halogenation reaction when it is treated with halogens. The addition of two halogen atoms takes place on the carbon-carbon double bond. The product formed is called as disubstituted
Trending nowThis is a popular solution!

Chapter 23 Solutions
OWLv2 6-Months Printed Access Card for Kotz/Treichel/Townsend's Chemistry & Chemical Reactivity, 9th, 9th Edition
- Give the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardWhich of the following is true of ethylene? a) It is not an important industrial chemical. b) It is present in large amounts in natural gas. c) It is formed by the catalytic hydrogenation of ethane. d) It is produced by heating ethane to around 800–900°C.arrow_forwardAre there cis/trans isomers of 1-propene?arrow_forward
- Draw the structure and give the molecular formula for a compound(a) 1-ethyl-3-methylcycloheptanearrow_forwardWrite two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures:(a) ethanol reacts with propionic acid(b) benzoic acid, C6H5CO2H, is added to a solution of sodium hydroxidearrow_forwardGive the general formula for any cyclic alkene, that is, acyclic hydrocarbon with one double bond.arrow_forward
- A certain hydrocarbon has a molecular formula of C5H8. Which of the following is not a structural possibility for this hydrocarbon? (a) It is a cycloalkane. (b) It contains one ring and one double bond. (c) It contains two double bonds and no rings. (d) It is an alkyne.arrow_forwardwhat is the structure pertaining to cis-1,3-dichlorocyclepentane?arrow_forwardWhat is the line structure and structural formula of 2-bromo-3-ethyl hexanal?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
