
Organic Chemistry, Loose-leaf Version
8th Edition
ISBN: 9781305865549
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 24, Problem 24.31P
Interpretation Introduction
Interpretation:
The possible reactants that would be placed in boxes in the sequence of Sonogashira coupling has to be given.
Concept Introduction:
Sonogashira coupling starts with a terminal alkyne with
This coupling is used to create diaryl
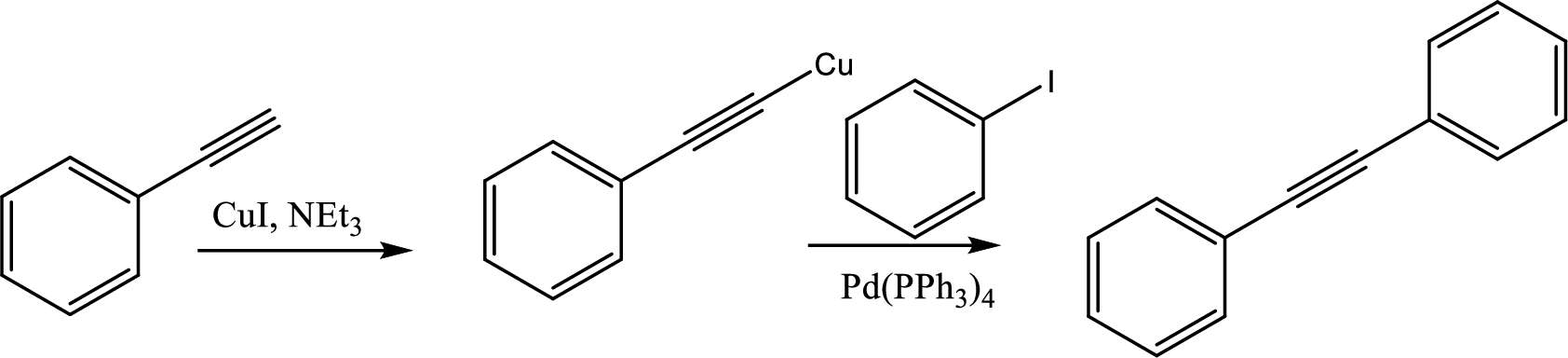
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The conversion of alcohols into alkyl halides by reaction with hydrogen halides is an example of a Nucleophilic Substitution Reaction. This kind of reaction can proceed by two different mechanisms depending on the structure of alcohol substrates used. Generally, primary alcohols are substituted via SN2 mechanism, while secondary and tertiary alcohols undergo SN1 mechanism.
Consider the following reaction given in the picture below and the questions in the picture too.
[Review Topics]
[References]
Acyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the
tetrahedral intermediate as it is first formed in the following reaction.
H3C
CH3
H2O
NaOH
• You do not have to consider stereochemistry.
• Include all valence lone pairs in your answer.
• Do not include counter-ions, e.g., Nat, I', in your answer.
• In cases where there is more than one answer, just draw one.
C
opy
aste
C.
Previous
Next
Email Instructor
Save and Ex
Cengage Learning | Cengage Technical Support
5:48 PI
82°F
3/28/20
Write a reaction that could be carried out to synthesize 1-(4-methylphenyl)ethanol (reactants will be a combination of Grignard reagent and carbonyl compound).
Chapter 24 Solutions
Organic Chemistry, Loose-leaf Version
Ch. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.4 - Prob. 24.3PCh. 24.5 - Show how the following compound can be prepared...Ch. 24.5 - Prob. 24.5PCh. 24.5 - Prob. 24.6PCh. 24.6 - Prob. 24.7PCh. 24 - Prob. 24.8PCh. 24 - Prob. 24.9PCh. 24 - Prob. 24.10P
Ch. 24 - Treatment of cyclohexene with iodobenzene under...Ch. 24 - Prob. 24.12PCh. 24 - Prob. 24.13PCh. 24 - The aryl diene undergoes sequential Heck reactions...Ch. 24 - Heck reactions take place with alkynes as well as...Ch. 24 - Prob. 24.16PCh. 24 - The following transformation involves a series of...Ch. 24 - Show the sequence of Heck reactions by which the...Ch. 24 - Prob. 24.19PCh. 24 - Write the steps that are critical in the following...Ch. 24 - Prob. 24.21PCh. 24 - Prob. 24.22PCh. 24 - Prob. 24.23PCh. 24 - Show how the following compound could be prepared...Ch. 24 - It is typically very difficult to do a...Ch. 24 - The compound eutypine is an antibacterial agent...Ch. 24 - Prob. 24.27PCh. 24 - Prob. 24.28PCh. 24 - Prob. 24.29PCh. 24 - Prob. 24.30PCh. 24 - Prob. 24.31PCh. 24 - Prob. 24.32PCh. 24 - Prob. 24.33PCh. 24 - The following transformation can be accomplished...Ch. 24 - Prob. 24.35PCh. 24 - Prob. 24.36PCh. 24 - Prob. 24.37PCh. 24 - Prob. 24.38PCh. 24 - E. J. Coreys 1964 total synthesis of...Ch. 24 - Prob. 24.40P
Knowledge Booster
Similar questions
- As presented in the chapter, the Sonogashira coupling reaction is commonly used to create diaryl alkynyl structures. However, it can also be used to create divinyl alkynyl structures. The following sequence of reactions creates such a product. Write possible reactants that would be placed in the boxes in the sequence. Si (Me)3 SiMe3 OSiMeg OSiMeg Pd(OAc), Cul, Et NH S-arrow_forwardPlease give the missing information like structure of reagents, products, etc..arrow_forwardThis reaction does not follow the expected theory. Given what you know about substitution/elimination, what is the expected mechanism this reaction should follow? Suggest why that is not what happens and give the actual product of the reaction. Br ткон sarrow_forward
- Supply the missing reagent for the following reactions.arrow_forwardWrite the products formed as a result of the reactions given below.arrow_forward[Review Topics] [References] Acyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the tetrahedral intermediate as it is first formed in the following reaction. %3D H3C CH3 HO-CH3 • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. • Do not include counter-ions, e.g., Na*, I', in your answer. • In cases where there is more than one answer, just draw one. C P. opy Bste C. Previous Next Email Instructor Save and Exit Cengage Learning | Cengage Technical Support 5:50 PM arch 82°F 3/28/2022arrow_forward
- 1. Draw the sulfur‑containing product of the oxidation reaction between two 2‑methyl‑1‑propanethiol molecules. Include all hydrogen atoms. 2. Modify the structure to draw the organic product of the following reaction. Note that [O] is present in excess. Modify the structure to represent the product.arrow_forwardArrange the following in order of decreasing reactivity to nucleophilic addition: ketone, aldehyde. Explain your logic.arrow_forwardWrite the product of the following sequence of reaction . how the combined reactions allow you to “navigate” between the different functional groupsarrow_forward
- Substitutions on aromatic rings are generally carried out by electrophilic substitution, but for the synthesis of phenolic compounds and alkoxy benzene it is carried out by nucleophilic substitution, explain why this is so .arrow_forwardDraw the structures of the two carbocation intermediates that might form during the reaction of propene (above) with HBr.arrow_forward1. Name and draw the structures of all the possible chemical (electrophilic aromatic substitution) reactions of acetophenone namely: * • Halogenation (chlorination or bromination) * • Nitration * • Sulphonation * • Friedel craft alkylation * • Friedel craft acylationarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning