
Concept explainers
(a)
Interpretation:
Whether hexose corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an
The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The sugar hexose is not the correct description of
Explanation of Solution
Monosaccharide with the six-carbon atom is known as hexose.
The presence of a keto group in
However, the keto group is missing in hexose as shown below.
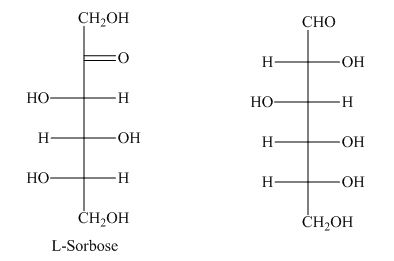
Figure 1
The term hexose is not the correct description of the
(b)
Interpretation:
Whether ketohexose corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The sugar ketohexose is the correct description of
Explanation of Solution
The class of the sugars which is fundamental in carbohydrates with presence of keto group is known as ketohexose sugars.
The presence of keto group in ketohexose shows similarity with
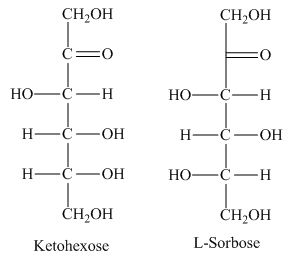
Figure 2
The ketohexose is the correct description of the
(c)
Interpretation:
Whether glycoside molecule corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The carbohydrate
Explanation of Solution
Two molecules of sugar which are connected to a glycosidic bond is known as glycoside molecule. The glycosidic bond is used to join two carbohydrate molecules.
The cyclic acetal group is not present in the
The glycoside molecule is not the correct description of the
(d)
Interpretation:
Whether aldohexose corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The carbohydrate
Explanation of Solution
The aldohexose belongs to the category of hexose in which aldehyde group is present at first carbon. In the
The sugar
(e)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is the correct description of
Explanation of Solution
The stereocenters are defined as the centers which are chiral in nature or attached with four different substituents. The stereocenters which are in direction of anticlockwise or left-hand nomenclaturehave
The configuration of
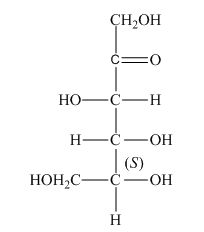
Figure 3
Therefore, the given structure is the correct description of
The given structure shown in Figure 3 is the correct description of
(f)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is not similar to the
Explanation of Solution
The stereocenters are defined as the centers which are chiral in nature or attached with four different substituents. The stereocenters which are in the direction of anticlockwise or left-hand nomenclature, the configuration of that stereocenter is
The configuration of
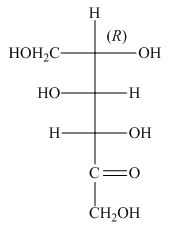
Figure 4
The given structure shown in Figure 4 is not the correct description of
(g)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is not similar to the
Explanation of Solution
The Haworth projection is used for the arrangements of cyclic sugars. The
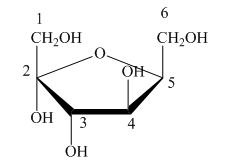
Figure 5
The given structure is not a proper description for
(h)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is not similar to the
Explanation of Solution
The Haworth projection is used for the arrangements of cyclic sugars. The
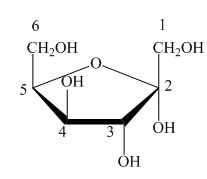
Figure 6
The configuration of sorbose is in
(i)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is similar to the
Explanation of Solution
The Haworth projection is used for the arrangements of cyclic sugars. The
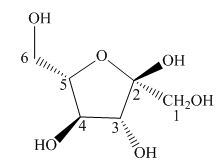
Figure 7
The given structure is the correct description for
(j)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
The pyranoses can be classified as
Answer to Problem 24.40AP
The given structure is not the correct description of
Explanation of Solution
The most stable conformation of cyclohexane is chair form due to the axial and equatorial position. The angle between the carbon-carbon bond is near about
The given structure is not the correct description of
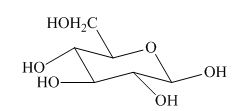
Figure 8
The given structure is not the correct description of
(k)
Interpretation:
Whether given structure corresponds to the correct description of the
Concept introduction:
Carbohydrates are a class of organic compounds. They can be present in the form of open chains or rings. They are usually an aldehyde or ketone with additional hydroxyl groups. The six-membered ring form of carbohydrates is termed as pyranose. The five-membered ring form of carbohydrates is termed as furanose.
Answer to Problem 24.40AP
The given structure is the correct description of
Explanation of Solution
The most stable conformation of cyclohexane is chair form due to the axial and equatorial position. The angle between the carbon-carbon bond is near about
All the substituents or groups are present in the same manner with respect to the
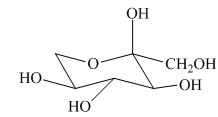
Figure 9
The given structure is the correct description of
Want to see more full solutions like this?
Chapter 24 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- Lactose is a disaccharide in which a glycosidic linkage connects the monosaccharides galactose and glucose. OH НО OH (a) Identify the glycosidic linkage and the acetal carbon in lactose. (b) What type of glycosidic linkage does lactose have (i.e., is it 1,1'-, 1,2'-, etc., and is it a or B)? (c) People who are lactose intolerant are deficient in the enzyme lactase, and therefore cannot efficiently break down the disaccharide into its monosaccharides. When lactose is treated with aqueous acid, however, this hydrolysis can take place, though relatively slowly. Draw the complete, detailed mechanism and the products of the acid-catalyzed hydrolysis of lactose. Но ОН НО ОН ОН Lactosearrow_forwardThe most stable conformation of the pyranose ring of most D-aldohexoses places the largest group, CH2OH, in the equatorial position. An exception to this is the aldohexose D-idose. Draw the two possible chair conformations of either the a or B anomer of D-idose. Explain why the more stable conformation has the CH2OH group in the axial position.arrow_forwardRegarding 4-O- (α-D-psychofuranosyl) -β-D-allopyranose. Please indicate the RIGHT alternative: (a) The disaccharide reacts with CH3OH in an acid medium to form a glycoside that cannot be oxidized with HNO3. (b) It is a reducing disaccharide only in basic medium. (c) In the structure there is only one glycosidic bond that is of the type β 1-O-4 ' (d) The hydrolysis products of this disaccharide do not show mutarrotation. (e) The disaccharide structure contains two six-membered rings.arrow_forward
- When a pyranose is in the chair conformation in which the CH2OH group and the C-1 OH group are both in axial positions, the two groups can react to form an acetal. This is called the anhydro form of the sugar (it has “lost water”). The anhydro form of D-idose is shown here. Explain why about 80% of d-idose exists in the anhydro form in an aqueous solution at 100 °C, but only about 0.1% of D-glucose exists in the anhydro form under the same conditions.arrow_forwardWhen a pyranose is in the chair conformation in which the CH2OH group and the C-1 OH group are both in axial positions, the two groups can react toform an acetal. This is called the anhydro form of the sugar (it has “lost water”). The anhydro form of d-idose is shown here. Explain why about 80% of d-idose exists in the anhydro form in an aqueous solution at 100 °C, but only about 0.1% of d-glucose exists in the anhydro form under the same conditions.arrow_forwardanswer correctly pleasearrow_forward
- Trehalose and maltose are both dimers of glucose. However, they have considereably different reactivities. Concisely explain why these differences are observed. но но HO HO но "HO он но он OH O HO OHOH но trehalose maltose 1. Malthose is a reducing sugar while trehalose is not. 2. Trehalose is very resistant to acid hydrolysis while maltose can be acid-hydrolyzed with ease.arrow_forwardConsider the structure of raffinose, a trisaccharide found in sugar beets and a number of higher plants. HO CH,OH Но- OH OCH, Но Но OH НОСН Но CH,OH ÓH raffinose (a) Classify raffinose as a reducing or nonreducing sugar, and tell how you know. (b) Identify the glycoside linkages in raffinose, and clas- sify each as either a or B. (c) Name the monosaccharides formed when raffinose is hydrolyzed in aqueous acid. (d) What products are formed when raffinose is treated with dimethyl sulfate in NaOH, and then with aqueous acid and heat?arrow_forward4. Identify the component monosaccharides of each of the following compounds and describe the type of glycosidic linkage in each. Но он Но OH HO он Но- Но- (a) OH (c) CH,OHO. (b) CH2OHO Lon OH HO H ČHOH H OH ÓH ОНarrow_forward
- C. Trehalose and maltose are both dimers of glucose. However, they have considereably different reactivities. Concisely explain why these differences are observed. HO НО НО HO Но HO OH Он HO OHOH Но trehalose maltose 1. Malthose is a reducing sugar while trehalose is not. 2. Trehalose is very resistant to acid hydrolysis while maltose can be acid-hydrolyzed with ease.arrow_forwardThe most stable conformation of most aldopyranoses is one in which the largest group, the CH2OH group, is equatorial. However, alpha-D-idopyranose exists primarily in a conformation with an axial CH2OH group. Write formulas for the two chair conformations of a-D-idopyranose (one with the CH2OH group axial and one with the CH2OH group equatorial) and provide an explanationarrow_forwardGive me a clear handwritten answer with explanationarrow_forward
