
Concept explainers
(a)
Interpretation:
The sequence of steps leading to the product and the role of sulfuric acid has to be shown.
Concept introduction:
Addition reaction:
Markovnikov’s rule:
In the overall reaction, the hydrogen atom (electrophile) is bonded to least substituted carbon and nucleophile is bonded to the most substituted carbon. This is known as Markovnikov’s rule.
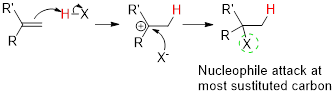
b)
Interpretation:
The structure of an alcohol that is an isomer of isopropanol has to be drawn.
Concept introduction:
Structural isomer: Molecules having same molecular formula but has different structural arrangements of atoms.
c)
Interpretation:
Chirality of isopropanol has to be interpreted.
Concept introduction:
Chirality: The presence of four different atoms (or groups) at carbon is known as asymmetric carbon and is known as chiral center of the compound.
d)
Interpretation:
The commercial use for the given compound 2-propanol has to be stated.
Concept introduction:
Functional groups:
Alcohol:
Commercial use of organic compounds: organic compounds are used as solvent, automobile fuel, lubricants, drugs, etc….
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Chemistry
- Acetyl chloride, CH3COCl, reacts with the hydroxyl groupsof alcohols to form ester groups with the elimination ofHCl. When an unknown compound X with formulaC4H8O3 reacted with acetyl chloride, a new compound Ywith formula C8H12O5 was formed.(a) How many hydroxyl groups were there in X?(b) Assume that X is an aldehyde. Write a possible structure for X and a possible structure for Y consistent with your structure for X.arrow_forwardAn isomer of C6H12O could contain a carboxylic acid. True or false?arrow_forwarddraw the structure of all monobromo derivatives of pentane, C5H11Br, which contain a 4 carbon chainarrow_forward
- Give the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forward(a) When a compound containing C, H, and O is completelycombusted in air, what reactant besides the hydrocarbonis involved in the reaction? (b) What products form in thisreaction? (c) What is the sum of the coefficients in the balancedchemical equation for the combustion of one mole ofacetone, C3H6O1l2, in air?arrow_forwardEnter the molecular formula for butane, C4H10?arrow_forward
- Write the chemical equation for the reaction of propanoic acid with 1-butanol (an alcohol). The formula of 1-butanol is CH-CH-CH2-CH2-OH. Which is formula of the this rule of reaction?arrow_forwardWhich of the following structural features could be found in a compound with formula C7H12?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning




