
Concept explainers
Interpretation:
The compound in the preceding steps has to be predicted.
Concept introduction:
Addition reaction:
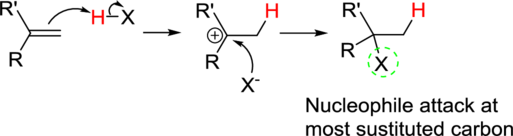
Markovnikov’s rule:
In the overall reaction, the hydrogen atom (electrophile) is bonded to least substituted carbon and nucleophile is bonded to the most substituted carbon. This is known as Markovnikov’s rule.
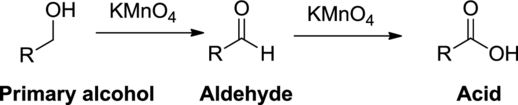
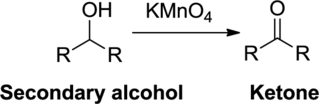
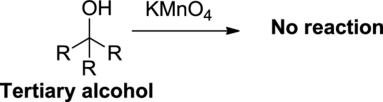
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidizing agent like
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Chemistry
- Compound A is unsaturated hydrocarbon with molecular formula (C6H12) reacted with Br2 in water to form compound B. compound C was produced from the reaction between compound A, sulphuric acid and H2O (g). Compound A undergo hydrogenation to form compound D. Compound E was produced from the reaction of compound A with Br2 in room temperature. Compound A undergo hydrohalogenation in the presences of hydrogen peroxide to form compound F. The reaction between compound F with aqueous sodium hydroxide will form compound G. Compound H was produced when compound F reacts with the aqueous ammonia in ethanol. Compound F also reacts with aqueous sodium cyanide to produce compound I. Draw the possible structural formulae of compounds A, B, C, D, E, F, G, H and I. Give the IUPAC nomenclature of compounds H and I. Distinguish between compound A and D.arrow_forward2-chloropropane is a major product of the reaction of chlorine with propane under ultraviolet light. Write the mechanism for this reaction including the initiation step and the two propagation steps.arrow_forwardAlkyl sulfonates undergo the same type of substitution reactions as alkyl halides and can also be prepared from alcohols. What advantage does the preparation of an alkyl sulfonate from an alcohol have over the preparation of an alkyl halide from an alcohol?arrow_forward
- One mole of an unknown hydrocarbon, compound C, in the presence of a platinum catalyst, adds 98.9 L of hydrogen, measured at 744 mm Hg and 22 degrees C , to form a saturated alkane which contains one ring. When one mole of compound C is reacted with ozone, followed by reduction with (CH3)2S , four moles of only one product was formed, whose condensed molecular formula is CHO -CHO. Give the structure of compound C. Explain your reasoningarrow_forwardThe acid-catalyzed dehydration of 2,3-dimethyl-3-pentanol yields three alkene products. What are the names of the three alkenes? Which of the three alkenes is the major product?arrow_forwardReaction of butane (CH 3CH 2CH 2CH 3) with Cl 2 in the presence of light forms two different alkyl chlorides that have molecular formula C 4H 9Cl. Draw the structures of both alkyl chlorides.arrow_forward
- Alkenes can be converted to alcohols by reaction with mercuric acetate to form a β-hydroxyalkylmercury(II) acetate compound, a reaction called oxymercuration. Subsequent reduction with NaBH4 reduces the C–Hg bond to a C–H bond, forming the alkyl alcohol, a reaction called demercuration. Draw the structures of the Hg-containing compound(s) and the final alcohol product(s) formed in the following reaction sequence, omitting byproducts. If applicable, draw hydrogen at a chirality center and indicate stereochemistry via wedge-and-dash bonds.arrow_forwardAlkenes can be converted to alcohols by reaction with mercuric acetate to form a β-hydroxyalkylmercury(II) acetate compound, a reaction called oxymercuration. Subsequent reduction with NaBH4 reduces the C–Hg bond to a C–H bond, forming the alkyl alcohol, a reaction called demercuration. Draw the structures of the Hg-containing compound(s) and the final alcohol product(s) formed in the following reaction sequence, omitting byproducts. If applicable, draw hydrogen at a chirality center and indicate stereochemistry via wedge-and-dash bonds.arrow_forwardCompound X (structure shown below) has a molecular formula C5H1o and reacts with H2/Pt to give compound Y, C5H12. What is the name of the reaction involved to produce Compound Y? H2C H3C CH3 Hydration Hydrogenation Halogenation Addition of halohydrinarrow_forward
- Hydrocarbon S, C8H8 reacts with hydrogen in the presence of platinum catalyst to yield T, C8H10. Compound U is formed when T is heated with alkaline potassium permanganate solution, followed by hydrolysis. The reaction of S with hydrogen bromide V, whereas ozonolysis of S produces W and methanal. Draw the structures of S to W. Give the name of the reaction for the conversion of S to T. Write all chemical equations involved.arrow_forwardThe major product formed when methylenecyclohexane is treated with NBS in dichloromethane is 1-(bromomethyl)cyclohexene. Account for the formation of this product.arrow_forwardThe reaction of Hbr with 2-methylpropene produces 2-bromo-2-methylpropane. What is the structure of the carbocation formed during the reaction?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





