
Concept explainers
Interpretation:
The structural formulas for the given compounds are to be provided.
Concept Introduction:
In Fischer projection formula, the horizontal and vertical line represents the bonds that are present above and below the plane, respectively.
The verticals bonds are represented as dashed wedge and horizontal bonds as dark wedge.
Answer to Problem 26P
Solution:
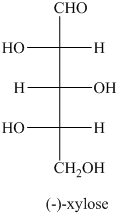
a) The structural formula of
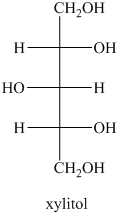
b) The structural formula for the xylitol is
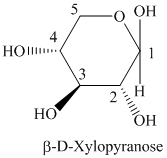
c) The structural formula of
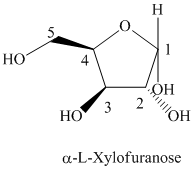
d) The structural formula of
e) The structure of
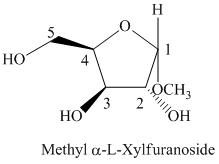
f) The Fischer projection formula for the given compound is shown below.
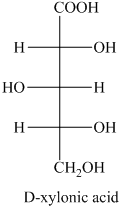
g) The structure of the given compound is shown below.
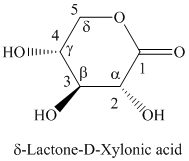
h) The structure of the given compound is shown below.
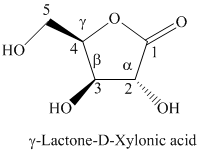
i) The Fischer projection formula for the given compound is shown below.
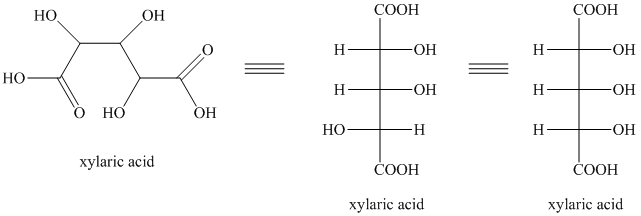
Explanation of Solution
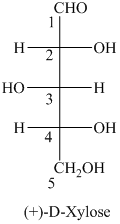
a) The given structure of
In,
L and D indicate the absolute configuration of a compound. If hydroxy group attached with chiral centre is to the right side and present on the bottom, then the carbohydrate is in D form but if hydroxy group is present to the left side then the carbohydrate is in L-form.
The name of the given compound is
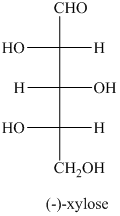
b) The given compound is xylitol.
The alcohol derivative of xylose is xylitol in which the reduction of

c) The given compound is
Sugar molecule that has six-membered-ring is known as pyranose and sugar molecule that has five-membered-ring is called furanose.
The given compound is formed when
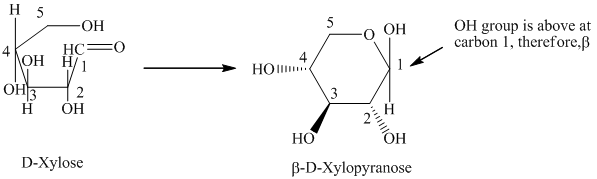
Thus, the structural formula of

d) The given compound is
L and D indicate the absolute configuration of a compound. If hydroxy group attached with chiral centre is to the right side and present on the bottom, then the carbohydrate is in D form but if hydroxy group is present to the left side then the carbohydrate is in L-form.
The given compound is formed when
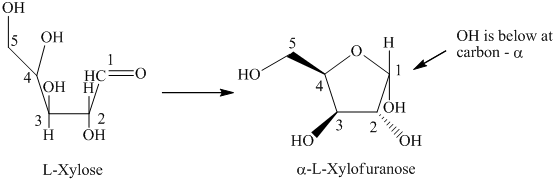
Thus, the structural formula of
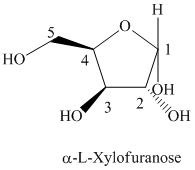
e) The given compound is
The given compound is formed when hydroxyl group is replaced by methoxy group as shown below.
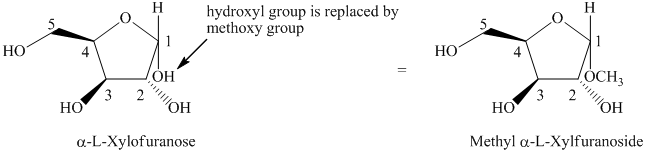
Thus, the structure of
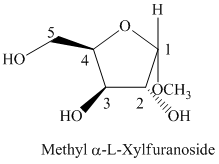
f) The given compound is
The oxidation of
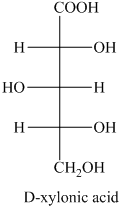
g) The given compound is
Lactones are class of organic compound which contain
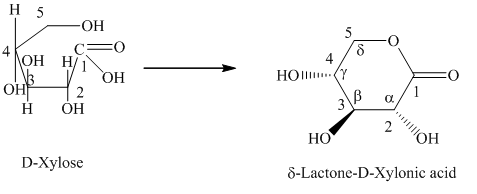
Thus, the structure of the given compound is shown below.

h) The given compound is
Lactones are class of organic compound which contain
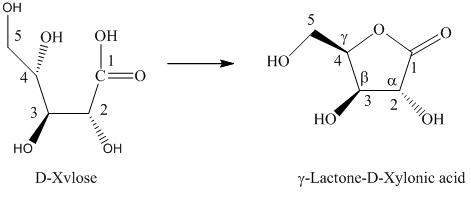
Thus, the structure of the given compound is shown below.
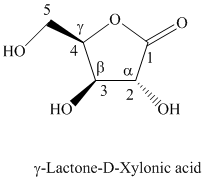
i) The given compound is Xylaric acid.
The oxidation of

Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- When oxaloacetic acid and acetyl-coenzyme A (acetyl-CoA) labeled with radioactive carbon-14 in position 2 are incubated with citrate synthase, an enzyme of the tricarboxylic acid cycle, only the following enantiomer of [2-l4C]citric acid is formed stereoselectively. Note that citric acid containing only 12C is achiral. Assign an R or S configuration to this enantiomer of [2-14C]citric acid. (Note: Carbon-14 has a higher priority than carbon-12.)arrow_forwardFollowing is a retrosynthesis for the coronary vasodilator ganglefene. (a) Propose a synthesis for ganglefene from 4-hydroxybenzoic acid and 3-methyl-3-buten-2-one. (b) Is ganglefene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardChoose the product that is expected when the β-pyranose form of compound A is treated with excess ethyl iodide in the presence of silver oxide. The following information can be used to determine the identity of compound A: 1. The molecular formula of compound A is C6H12O6.2. Compound A is a reducing sugar.3. When compound A is subjected to a Wohl degradation two times sequentially, D-erythrose is obtained.4. Compound A is epimeric with D-glucose at C3.5. The configuration at C2 is R.arrow_forward
- Hyaluronic acid, a component of connective tissue, is the fluid that lubricates joints. It is a polymer of alternating N-acetyl-d-glucosamine and d-glucuronic acid subunits joined by β-1,3'-glycosidic linkages. Draw a short segment of hyaluronic acid.arrow_forwardEmil Fischer synthesized l-gulose, an unusual aldohexose that reduces to give d-glucitol.Suggest a structure for this l sugar, and show how l-gulose gives the same alditol asd-glucose. (Hint: d-Glucitol has ¬CH2OH groups at both ends. Either of these primaryalcohol groups might have come from reduction of an aldehyde.)arrow_forwardMannose is an aldohexose with relative stereochemistry AABB. i) Draw the Fischer projection of 2-amino-2-deoxy-D-mannose. ii) Construct the Haworth projection of 2-amino-2-deoxy-D-mannose. iii) Assign the absolute configuration for C-3 in 2-amino-2-deoxy-D-mannose.arrow_forward
- Is meclizine chiral? If so, how many of the possible stereoisomers are formed in this synthesis?arrow_forwardDraw 1D 1H NMR spectrum of Glycine1) in Deuterated chloroform CDCl3 and 2) in Heavy Water D2Oarrow_forwardAn optically active D-aldopentose (A) produced an optically inactive alditol (B) upon treatment with H2/Pt. When the aldopentose (A) was subjected to a Ruff degradation, D-aldotetrose (C) was generated. The aldotetrose (C) gave an optically active aldaric acid (D) upon oxidation with HNO3. D-aldopentose (A) can be prepared from D-threose by a Killani Fischer synthesis. Propose structure of A through D.arrow_forward
- (a) Which of the d-aldopentoses will give optically active aldaric acids on oxidation with HNO3 ?(b) Which of the d-aldotetroses will give optically active aldaric acids on oxidation with HNO3 ?(c) Sugar X is known to be a d-aldohexose. On oxidation with HNO3, X gives an optically inactive aldaric acid. WhenX is degraded to an aldopentose, oxidation of the aldopentose gives an optically active aldaric acid. Determine thestructure of X.(d) Even though sugar X gives an optically inactive aldaric acid, the pentose formed by degradation gives an opticallyactive aldaric acid. Does this finding contradict the principle that optically inactive reagents cannot form opticallyactive products?(e) Show what product results if the aldopentose formed from degradation of X is further degraded to an aldotetrose.Does HNO3 oxidize this aldotetrose to an optically active aldaric acid?arrow_forwardWhat aldoses are formed when the following aldoses are subjected to the Kiliani–Fischer synthesis: (a) d-threose; (b) dribose; (c) D-galactose?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


