
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24.4, Problem 11P
What reagents are needed to convert 2-methylpropene
a.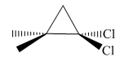 b.
b. 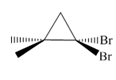 c.
c. 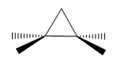
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: [1] NaBH4, CH3OH; [2] CH3MgBr, then H2O; [3] Ph3P = CHOCH3; [4] CH3CH2CH2NH2, mild acid; [5] HOCH2CH2CH2OH, H+.
a) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers that are not tautomers, but contain a C = C and an OH group.
Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation.
c. CH3CH2Cl, AlCl3
l. product in (c), then KMnO4
Chapter 24 Solutions
Organic Chemistry (6th Edition)
Ch. 24.1 - Prob. 1PCh. 24.1 - Prob. 2PCh. 24.1 - Prob. 3PCh. 24.2 - Prob. 4PCh. 24.2 - Prob. 5PCh. 24.4 - Problem 26.10
What reagents are needed to convert...Ch. 24.5 - Problem 26.11
What product is formed when each...Ch. 24.5 - Prob. 13PCh. 24.6 - Problem 26.13
Draw the products formed when each...Ch. 24.6 - Problem 26.14
What products are formed when ...
Ch. 24 - 26.19 What product is formed by ring-closing...Ch. 24 - 26.20 Draw the products formed in each...Ch. 24 - What organic halide is needed to convert lithium...Ch. 24 - 26.22 How can you convert ethynylcyclohexane to...Ch. 24 - 26.23 What compound is needed to convert styrene...Ch. 24 - 26.24 What steps are needed to convert to octane...Ch. 24 - Prob. 27PCh. 24 - 26.27 Draw the products (including stereoisomers)...Ch. 24 - 26.28 Treatment of cyclohexene with and forms...Ch. 24 - Prob. 32PCh. 24 - 26.30 What starting material is needed to prepare...Ch. 24 - Prob. 37PCh. 24 - Prob. 38PCh. 24 - When certain cycloalkenes are used in metathesis...Ch. 24 - 26.34 Draw the products formed in each reaction.
...Ch. 24 - Prob. 41PCh. 24 - Draw a stepwise mechanism for the following...Ch. 24 - Sulfur ylides, like the phosphorus ylides of...Ch. 24 - Although diazomethane is often not a useful...Ch. 24 - Prob. 46PCh. 24 - Prob. 47PCh. 24 - 26.45 Devise a synthesis of each compound from...Ch. 24 - 26.46 Devise a synthesis of each substituted...Ch. 24 - Biaryls, compounds containing two aromatic rings...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the product formed when (CH3)2CHOH is treated with each reagent (d, e and f)arrow_forwardDraw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each Wittig reagent.arrow_forwardDraw the product formed when C6H5N2 +Cl− reacts with each compound.arrow_forward
- Draw the product formed when C6H5N2+Cl− reacts with each compound.arrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forwardDraw the products formed when A is treated with each reagent: (a) H2 + Pd-C; (b) mCPBA; (c) PCC; (d) CrO3, H3SO4, H2O; (e) Sharpless reagent with (+)-DET.arrow_forward
- Determine the product formed when compound A is treated with each reagent.arrow_forwardA is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that exhibits stereoisomerism as E or Z.arrow_forward(a) What is the major alkene formed when A is dehydrated with HS2O4? (b) What is the major alkene formed when A is treated with POCl3 and pyridine? Explain why the major product is different in these reactions.arrow_forward
- Draw the product formed when (CH3)2CHOH is treated with each reagent. a.SOCl2, pyridine b. TsCl, pyridine c.H2SO4 d.HBr e.PBr3, then NaCN f.POCl3, pyridinearrow_forwardDraw the product formed when (CH3)2CHOH is treated with each reagent. a. SOCl2, pyridine b. TsCl, pyridine c. H2SO4 d. HBr e. PBr3, then NaCN f. POCl3, pyridinearrow_forwardAnswer each question using the ball-and-stick model of compound A. Draw a constitutional isomer that contains an ether and give its IUPACname. Draw the products formed (including stereochemistry) when A istreated with each reagent: [1] NaH; [2] H2SO4; [3] POCl3, pyridine; [4]HCl; [5] SOCl2, pyridine; [6] TsCl, pyridinearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY