
Interpretation:
The major products that are formed from the given organic reaction are to be determined. The stereochemistry that exhibits by the diastereomeric products, if they are formed by these reactions is to be explained.
Concept Introduction:
The catalytic hydrogenation reaction is an addition reaction of hydrogen gas in a molecule in the presence of a catalyst.
Answer to Problem 22P
Solution:
a) The principal organic product is.:
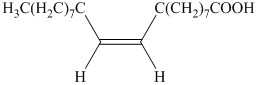
b) The principal organic product is:
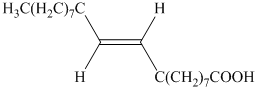
c) The principal organic product is:
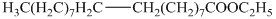
d) The principal organic product is:
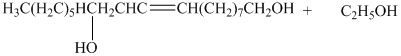
e) The principal organic product is:
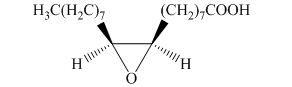
f) The principal organic product is:
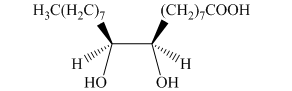
g) The principal organic product is:
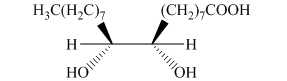
h) The principal organic product is:
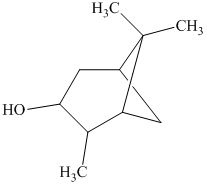
i) The principal organic product is:
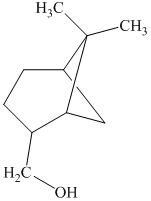
j) The principal organic product is:
Explanation of Solution
a) The given organic reaction isshown below.
The given reaction is the hydrogenation of
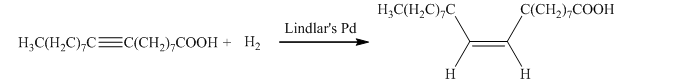
b) The given organic reaction is shown below.
The given reaction is the hydrogenation of alkynes in the presence of
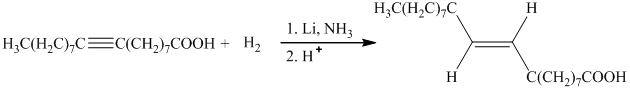
c) The given organic reaction is shown below.
The given reaction is the hydrogenation of alkenes in the presence of a platinum catalyst that reducesalkenes to
d) The given organic reaction is shown below.
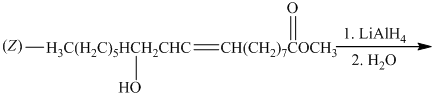
The carboxylic acid and its derivatives are reduced to the primary alcohol with the help of strong reducing agents such as lithium aluminum hydride,
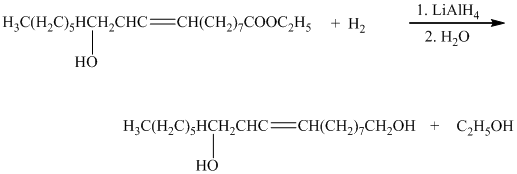
e) The given organic reaction is shown below.
In the given reaction, the epoxidation of alkene takes place. In such a reaction, the transfer of an oxygen atom from the peracid to
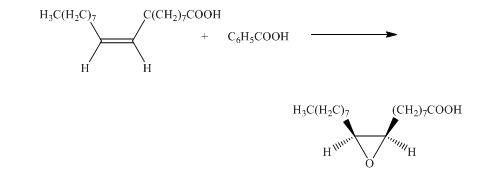
f) The given organic reaction is shown below.
In the given reaction, the hydrolysis of the product takes place that results in the breaking of the oxirane ring and forms a vicinal diol that is associated with two hydroxyl groups present on adjacent carbon atoms. The principal organic product of the given reaction is shown below.
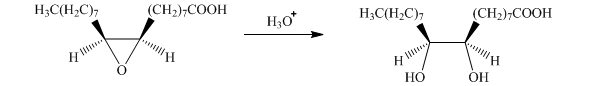
g) The given organic reaction is shown below.
In the given reaction, osmium tetraoxide
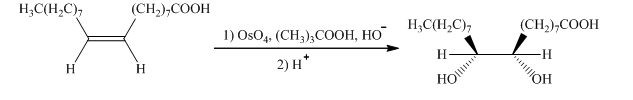
h) The given organic reaction is shown below.
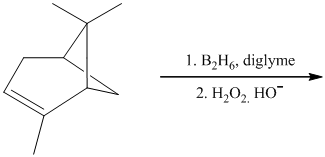
In the given reaction, hydroboration of alkenes takes place with the help of
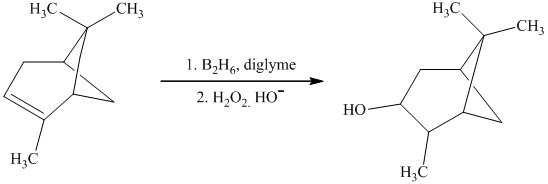
i) The given organic reaction is shown below.
In the given reaction, hydroboration of alkenes taken place with the help of
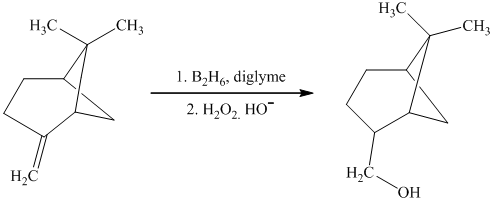
j) The given organic reaction is shown below.
In the given reaction, the dehydration of secondary alcohol is taken place in the presence of
Want to see more full solutions like this?
Chapter 25 Solutions
ORGANIC CHEM BUNDLE >BI<
- Terpin, prepared commercially by the acid-catalyzed hydration of limonene, is used medicinally as an expectorant for coughs. (a) Propose a structural formula for terpin and a mechanism for its formation. (b) How many cis, trans isomers are possible for the structural formula you propose?arrow_forward7. When butan-2-ol undergoes acid catalysed dehydration reaction, a mixture of products, B and C, are formed.Compound C shows cis-trans isomerism. Ozonolysis of compound B yields D and E. Draw the structure of Band C, and determine the major product. Draw cis-trans isomers of C. Write the reaction equation for theozonolysis of compound B that yields D and E. Show the preparation of butan-2-ol via a Grignard reagent byusing a suitable alkyl halide and compound D and E.arrow_forwardThe optically active (2R)-2-phenyl-2-butanol reacts in hydrochloric acid to form haloalkanes and alkenes. The substitution reaction is reported to occur with 100% racemization. Give the structures of the enantiomers that form during the substitution and indicate how much of each is formed. Propose a mechanism for the substitution reaction to yield the R product. What reagents would you use to achieve 100% retention of configuration. Two geometric isomers are obtained during the elimination reaction. Explain mechanistically which alkene will be the main product.arrow_forward
- Compound X is optically inactive and has the formula C 16H 16Br 2. On treatment with strong base, X gives hydrocarbon Y, C 16H 14. Compound Y absorbs 2 equivalents of hydrogen when reduced over a palladium catalyst and reacts with ozone to give two fragments. One fragment Z, is an aldehyde with formula C 7H 6O. The other fragment is glyoxal, (CHO)2. Which of the following answers is correct? Select all that are correct.arrow_forward5. Compound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct.arrow_forwardCompound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct. Can be more than one answerarrow_forward
- Which of the following results would we look for if we were to distinguish the two compounds below by addition of the mentioned reagents/subjecting the compounds to the reactions? a. Decolorization of KMnO4 b. Decolorization of Br2 in light c. Decolorization of AlCl3 when reacted with the compound and Br2 d. Yellow, sooty flame under combustionarrow_forwardOn acid-catalyzed dehydration, 1-butanol (CH3CH2CH2CH2OH) can be converted to 1-butene. Write out an equation for the reaction Assign each the appropriate symbol for the mechanism of the reaction (E1 or E2) Draw a suitable mechanism for the reactionarrow_forwardIgnoring stereochemistry, draw the alkylborane formed from the addition of one equivalent of BH3 to the alkene. The alkylborane formed in Part 1 is further treated with H2O2 and HO−. Draw the two stereoisomers of the final product of the reaction. Include stereochemistry where relevant. How many stereocenters are formed from the reaction? What is the relationship between the stereoisomers?arrow_forward
- Three isomeric pentanols with unbranched carbon chains exist. Which of these isomers, upon dehydration at 180C, yields only 1-pentene as a product?arrow_forwardThe following bicyclic ketone has two -carbons and three -hydrogens. When this molecule is treated with D2O in the presence of an acid catalyst, only two of the three -hydrogens exchange with deuterium. The -hydrogen at the bridgehead does not exchange. How do you account for the fact that two -hydrogens do exchange but the third does not? You will find it helpful to build models of the enols by which exchange of -hydrogens occurs.arrow_forwardWhat is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at room temperature? Disregard stereoisomers.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,



