
ORGANIC CHEMISTRY W/CONNECT
10th Edition
ISBN: 9781307048513
Author: Carey
Publisher: MCG/CREATE
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25, Problem 29P
A synthesis of triacylglycerols has been described that begins with the substance shown.
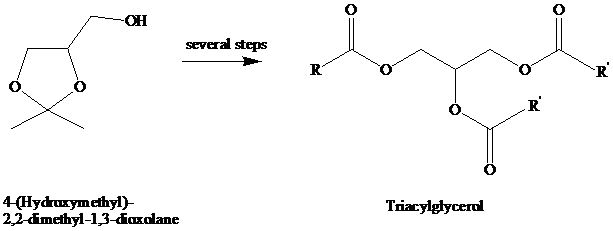
Outline a series of reactions suitable for the preparation of a triacylglycerol of the type
illustrated in the equation, where R and R′ are different.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
State the type of reaction and give the structure of the product when cholesterol is heated with ethanoic acid ( CH3COOH) in the presence of concentrated sulphuric acid as catalyst.
Which of the following statements is incorrect?
A. Because like dissolves like, the nonpolar end of the soap molecule can dissolve greasy dirt, and the polar or ionic end of the molecule is attracted by water molecules.
B. By saturating the soap solution with NaCl or KCl, the saturation products of the Na+ and K+ salts of fatty acids are exceeded and most of the soap is precipitated, while glycerin dissolves in water.
The alkalinity provided by C. HO- damages the leather, making it difficult to remove dirt.D. Soaps are divided into two as hard and soft according to their consistency. Saturated and single-double-bonded fatty acids are used in hard soap making. Mild soap is obtained from fatty acids containing two or three double bonds.
explain further why this statement is true
"A saturated fatty acid sample has a high boiling point. A triacylglycerol is composed of glycerol and three saturated fatty acids. Therefore, a sample not containing this triacylglycerol will have a low boiling point."
Chapter 25 Solutions
ORGANIC CHEMISTRY W/CONNECT
Ch. 25.2 - What fatty acids are produced on hydrolysis of...Ch. 25.3 - Give the structure of the keto acyl-ACP formed...Ch. 25.4 - Prob. 3PCh. 25.4 - Prob. 4PCh. 25.5 - Prob. 5PCh. 25.6 - Write the structural formula and give the IUPAC...Ch. 25.6 - Prob. 7PCh. 25.7 - Locate the isoprene units in each of the...Ch. 25.7 - Prob. 9PCh. 25.9 - Prob. 10P
Ch. 25.9 - Prob. 11PCh. 25.10 - Prob. 12PCh. 25.11 - Prob. 13PCh. 25.11 - Prob. 14PCh. 25.12 - Prob. 15PCh. 25.16 - Prob. 16PCh. 25 - Prob. 17PCh. 25 - Prob. 18PCh. 25 - Prob. 19PCh. 25 - Pyrethrins are a group of naturally occurring...Ch. 25 - Prob. 21PCh. 25 - Prob. 22PCh. 25 - Prob. 23PCh. 25 - Prob. 24PCh. 25 - Prob. 25PCh. 25 - Prob. 26PCh. 25 - Prob. 27PCh. 25 - Prob. 28PCh. 25 - A synthesis of triacylglycerols has been described...Ch. 25 - Prob. 30PCh. 25 - Prob. 31PCh. 25 - Prob. 32PCh. 25 - Prob. 33DSPCh. 25 - Prob. 34DSPCh. 25 - Prob. 35DSPCh. 25 - Prob. 36DSPCh. 25 - Prob. 37DSPCh. 25 - Prob. 38DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using your own words and illustrations, concisely discuss the absorption of triglycerides, phospholipids, and cholesterol esters from food. Give answer all questions with explanation.arrow_forwardHow many different triacylglycerols are there in which one of the fatty acid components is lauric acid, one is myristic acid, and one is palmitic acid?arrow_forwardEnumerate the appropriate methods of extraction and purification of glycosides.arrow_forward
- Which fatty acid is likely to occur commonly in natural sources?arrow_forwardThere are two isomeric triacylglycerol molecules whose components are glycerol, one palmitic acid unit, and two stearic acid units. Draw the structures of both, and explain how they differ.arrow_forwardWhich of the following statements is incorrect? A) Because like dissolves like, the nonpolar end of the soap molecule can dissolve greasy dirt, and the polar or ionic end of the molecule is attracted by water molecules. B) By saturating the soap solution with NaCl or KCl, the saturation products of the Na+ and K+ salts of fatty acids are exceeded, and most of the soap is precipitated, while glycerin dissolves in water. C) The alkalinity provided by HO- damages the leather, thus making it difficult to remove the dirt. D) Soaps are divided into two as hard and soft according to their consistency. Saturated and single-double-bonded fatty acids are used in soap making. Mild soap is obtained from fatty acids containing two or three double bonds.arrow_forward
- this statement is true, please prvide counterargument "A saturated fatty acid sample has a high boiling point. A triacylglycerol is composed of glycerol and three saturated fatty acids. Therefore, a sample not containing this triacylglycerol will have a low boiling point."arrow_forwardprovide factual evidence that this is true "A saturated fatty acid sample has a high boiling point. A triacylglycerol is composed of glycerol and three saturated fatty acids. Therefore, a sample not containing this triacylglycerol will have a low boiling point."arrow_forwardDescribe the formation of hydroxyproline for the synthesis of collagen in detailarrow_forward
- Biodiesel, a fuel typically derived from plant oils, can be manufactured by treating the oil with a methanol/KOH mixture to produce fatty acid methyl esters. Indicate the structures of the products generated by treating the triacylglycerol with methanol/KOH.arrow_forward(a) A certain lipid has the structure shown below (R1 and R2 are fatty acyl chains). Could a sample of these molecules form a bilayer? Why or why not? (b) Would phospholipase A1 be able to use this molecule as a substrate? Why or why not?arrow_forwardIs this fatty acid saturate, monounsaturate, polyunsaturate or none of the choices shown?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY