
ORGANIC CHEMISTRY (LL)-W/MOD.MASTERING.
8th Edition
ISBN: 9780134466712
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25.8, Problem 20P
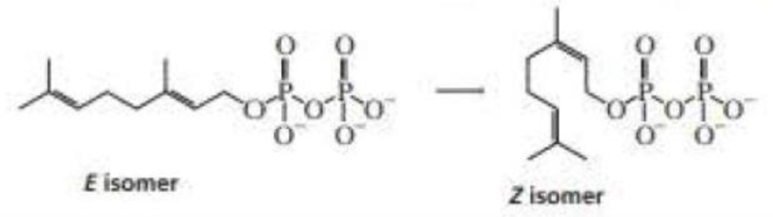
Propose a mechanism for the conversion of the E isomer of geranyl pyrophosphate to the Z isomer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the products of the addition of -OH to the si face of crotonate at C3, followed by protonation at C2, also from the si face
Explain the Mechanism - Isomerization of Geranyl Diphosphate to NerylDiphosphate ?
Propose a mechanism for base-promoted conversion of the proposed intermediate to sodium cyclopentanecarboxylate
Chapter 25 Solutions
ORGANIC CHEMISTRY (LL)-W/MOD.MASTERING.
Ch. 25.1 - Prob. 1PCh. 25.3 - Which has a higher melting point, glyceryl...Ch. 25.3 - Draw the structure of an optically inactive fat...Ch. 25.3 - Draw the structure of an optically active fat...Ch. 25.5 - Do the identities of R1 and R2 in phosphatidic...Ch. 25.5 - Membranes contain proteins, Integral membrane...Ch. 25.5 - Prob. 8PCh. 25.5 - The membrane phospholipids in deer have a higher...Ch. 25.6 - Treating PGC2 with a strong base such as sodium...Ch. 25.7 - Mark off the isoprene units in menthol, -selinene,...
Ch. 25.7 - Prob. 13PCh. 25.7 - Prob. 14PCh. 25.8 - Propose mechanisms for the Claisen condensation...Ch. 25.8 - Prob. 16PCh. 25.8 - Propose a mechanism for the conversion of...Ch. 25.8 - Propose a mechanism for the biosynthesis of...Ch. 25.8 - Propose a mechanism for the conversion of the E...Ch. 25.8 - The fluoro-substitued geranyl pyrophosphate shown...Ch. 25.8 - Prob. 22PCh. 25.8 - Prob. 23PCh. 25.9 - Draw the individual 1,2-hydride and 1,2-methyl...Ch. 25.10 - Prob. 26PCh. 25.10 - Prob. 27PCh. 25.10 - The acid component of a cholesterol ester is a...Ch. 25.10 - Prob. 29PCh. 25.10 - Prob. 30PCh. 25 - Prob. 31PCh. 25 - An optically active fat, when completely...Ch. 25 - Prob. 33PCh. 25 - a. How many different triacylglycerols are there...Ch. 25 - Cardiolipins are found in heart muscles. Draw the...Ch. 25 - Nutmeg contains a simple, fully saturated...Ch. 25 - Draw the product that is obtained from the...Ch. 25 - Prob. 39PCh. 25 - Prob. 40PCh. 25 - Propose a mechanism for the biosynthesis of...Ch. 25 - 5-Androstene-3.17-dione is isomerized to...Ch. 25 - Prob. 44PCh. 25 - Eudesmol is a sesquiterpene found in eucalyptus....Ch. 25 - Prob. 46PCh. 25 - Prob. 47PCh. 25 - Diethylstilbestrol (DES) was given to pregnant...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Does the reduction of acetoacetyl ACP in step 6 occur on the Re face or the Si face of the molecule?arrow_forwardOzonolysis, osmylation, and epoxide formation with mCPBA are all what type of reactions? a.) reduction b.)oxidation c) bothd) neitherarrow_forwardE53. why does alkaline phosphatase optimal at pH of 8 rather than pH 7arrow_forward
- 3. Write the mechanism for the formation of cychlohexanone oxime.arrow_forwardWrite a mechanism for the bromination of trans-cinnamic acid that proceeds through a bromonium ion intermediate that clearly shows the stereochemical outcome of the reaction. Show (R,S) configurations of the product(s).arrow_forwardThe reaction for the magnesium cation with 8-hydroxyquinoline is carried out in the presence of: A) Nitric acid B) Sodium hydroxide C) A solution of ammonia and ammonium chloride D) Sulfuric acid E) Sodium acetatearrow_forward
- State various conditions required in preparation of H2SO4?arrow_forwardA strong acid is used to catalyze this Fischer Esterification of acetic acid. What two steps in this reaction are accelerated by the presence of strong acid, and what function does the acid play in each of these steps?arrow_forwardPropose a mechanism for the conversion of mevalonic acid to mevalonyl pyrophosphate.arrow_forward
- Triosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme’s catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a general-base and a general-acid catalyst, respectively. Propose a mechanism for the reaction.arrow_forwardProvide the structures of intermediates J, L and N in the answer boxes provided.arrow_forwardA coupling reaction between lithium dimethylcuprate and an organobromide produces the compounds shown. Draw the structure of the most efficient organobromide reactant, assuming monoalkylation occurs.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License