
ORGANIC CHEMISTRY-MOLYMOD PACKAGE
5th Edition
ISBN: 9781260227307
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 26, Problem 26.11P
What product is formed when each
a.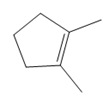 b.
b. 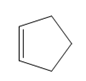 c.
c. 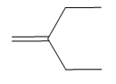
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the product formed when (CH3)2CHOH is treated with each reagent (d, e and f)
Draw the products formed when A is treated with each reagent: (a) H2 + Pd-C; (b) mCPBA; (c) PCC; (d) CrO3, H3SO4, H2O; (e) Sharpless reagent with (+)-DET.
What product is formed when each alkene is treated with CH2I2 and Zn(Cu)?
Chapter 26 Solutions
ORGANIC CHEMISTRY-MOLYMOD PACKAGE
Ch. 26 - Prob. 26.1PCh. 26 - Prob. 26.2PCh. 26 - Prob. 26.3PCh. 26 - Prob. 26.4PCh. 26 - Prob. 26.5PCh. 26 - Prob. 26.6PCh. 26 - Prob. 26.7PCh. 26 - Problem 26.8
What starting materials are needed to...Ch. 26 - Prob. 26.9PCh. 26 - Problem 26.10
What reagents are needed to convert...
Ch. 26 - Problem 26.11
What product is formed when each...Ch. 26 - Prob. 26.12PCh. 26 - Problem 26.13
Draw the products formed when each...Ch. 26 - Problem 26.14
What products are formed when ...Ch. 26 - Problem 26.15
Draw the product formed from...Ch. 26 -
What product is formed by ring-closing metathesis...Ch. 26 - Problem 26.17
What starting material is needed to...Ch. 26 - 26.18 In addition to organic halides, alkyl...Ch. 26 - 26.19 What product is formed by ring-closing...Ch. 26 - 26.20 Draw the products formed in each...Ch. 26 - What organic halide is needed to convert lithium...Ch. 26 - 26.22 How can you convert ethynylcyclohexane to...Ch. 26 - 26.23 What compound is needed to convert styrene...Ch. 26 - 26.24 What steps are needed to convert to octane...Ch. 26 - Prob. 26.25PCh. 26 - Prob. 26.26PCh. 26 - 26.27 Draw the products (including stereoisomers)...Ch. 26 - 26.28 Treatment of cyclohexene with and forms...Ch. 26 - Prob. 26.29PCh. 26 - 26.30 What starting material is needed to prepare...Ch. 26 - Prob. 26.31PCh. 26 - Prob. 26.32PCh. 26 - When certain cycloalkenes are used in metathesis...Ch. 26 - 26.34 Draw the products formed in each reaction.
...Ch. 26 - Prob. 26.35PCh. 26 - Draw a stepwise mechanism for the following...Ch. 26 - Sulfur ylides, like the phosphorus ylides of...Ch. 26 - Although diazomethane is often not a useful...Ch. 26 - Prob. 26.39PCh. 26 - Prob. 26.40PCh. 26 - Prob. 26.41PCh. 26 - Prob. 26.42PCh. 26 - 26.43 Devise a synthesis of each compound using a...Ch. 26 - 26.44 Devise a synthesis of each compound from...Ch. 26 - 26.45 Devise a synthesis of each compound from...Ch. 26 - 26.46 Devise a synthesis of each substituted...Ch. 26 - Biaryls, compounds containing two aromatic rings...Ch. 26 - Prob. 26.48PCh. 26 - 26.49 Draw the product formed from the...Ch. 26 - Prob. 26.50PCh. 26 - 26.51 Devise a synthesis of each of the following...Ch. 26 - Prob. 26.52PCh. 26 - 26.53 The following conversion, carried out in the...Ch. 26 - Prob. 26.54PCh. 26 - 26.55 Dimethyl cyclopropanes can be prepared by...Ch. 26 - Prob. 26.56P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each Wittig reagent.arrow_forward(a) Give the IUPAC name for A and B. (b) Draw the product formed when A or B is treated with each reagent: [1] NaBH4, CH3OH; [2] CH3MgBr, then H2O; [3] Ph3P = CHOCH3; [4] CH3CH2CH2NH2, mild acid; [5] HOCH2CH2CH2OH, H+.arrow_forwardWhat product is formed when each alkene is treated with HCl?arrow_forward
- a) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers that are not tautomers, but contain a C = C and an OH group.arrow_forwardDimethyl cyclopropanes can be prepared by the reaction of an α,β-unsaturated carbonyl compound X with two equivalents of a Wittig reagent Y. Draw a stepwise mechanism for this reaction.arrow_forwardDimethyl cyclopropanes can be prepared by the reaction of an α, β- unsaturated carbonyl compound X with two equivalents of a Wittig reagent Y. Draw a stepwise mechanism for this reaction.arrow_forward
- Determine the product formed when compound A is treated with each reagent.arrow_forwardDraw the products formed when (CH3)2C=CH2 is treated with following reagent. Cl2arrow_forwardDraw the product formed when (CH3)2CHOH is treated with each reagent. a.SOCl2, pyridine b. TsCl, pyridine c.H2SO4 d.HBr e.PBr3, then NaCN f.POCl3, pyridinearrow_forward
- Draw the product formed when (CH3)2CHOH is treated with each reagent. a. SOCl2, pyridine b. TsCl, pyridine c. H2SO4 d. HBr e. PBr3, then NaCN f. POCl3, pyridinearrow_forwardDraw the organic products formed when cyclopentene is treated withfollowing reagent. CH3CO3Harrow_forwardDraw all constitutional isomers formed when each alkene is treated with NBS + hv.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY