
Organic Chemistry, Loose-leaf Version
8th Edition
ISBN: 9781305865549
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 26, Problem 26.20P
Interpretation Introduction
Interpretation:
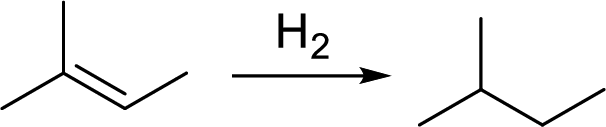
The structural formula of product of treatment of cholesterol with
Concept Introduction:
Catalytic hydrogenation: It is the addition of molecular hydrogen in double bond in presence of poisoned metal catalyst .
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Define Glycoside Hydrolysis ?
Draw the structural formula for the product formed by treatment of cholesterol with H2/Pd; then do the same for Br2
است
Take a look at this molecule, and then answer the questions in the table below it.
H
OH
CH₂OH
H
O
OH OH
H
H
H
O
CH₂OH
H
OH H
H N
H
O
Is this a reducing sugar?
H
L OH
OH
Does this molecule contain a glycosidic bond?
If you said this molecule does contain a glycosidic bond, write the symbol
describing it.
If you said this molecule does contain a glycosidic bond, write the common
names (including anomer and enantiomer labels) of the molecules that
would be released if that bond were hydrolyzed.
If there's more than one molecule, separate each name with a comma.
O yes
O no
O yes
O no
0-0
0
α
X
В
ローロ
S
Chapter 26 Solutions
Organic Chemistry, Loose-leaf Version
Ch. 26.1 - Prob. 26.1PCh. 26.6 - Prob. AQCh. 26.6 - Prob. BQCh. 26.6 - Prob. CQCh. 26.6 - Prob. DQCh. 26.6 - Prob. FQCh. 26 - Prob. 26.2PCh. 26 - Identify the hydrophobic and hydrophilic region(s)...Ch. 26 - Prob. 26.4PCh. 26 - Prob. 26.5P
Ch. 26 - Prob. 26.6PCh. 26 - Prob. 26.7PCh. 26 - Prob. 26.8PCh. 26 - Prob. 26.9PCh. 26 - How many moles of H2 are used in the catalytic...Ch. 26 - Prob. 26.11PCh. 26 - Prob. 26.12PCh. 26 - Prob. 26.13PCh. 26 - Prob. 26.14PCh. 26 - Prob. 26.15PCh. 26 - Prob. 26.16PCh. 26 - Prob. 26.17PCh. 26 - Prob. 26.18PCh. 26 - Prob. 26.20PCh. 26 - Prob. 26.21PCh. 26 - Prob. 26.22PCh. 26 - Prob. 26.23PCh. 26 - Following is a structural formula for cortisol...Ch. 26 - Prob. 26.25PCh. 26 - Draw the structural formula of a lecithin...Ch. 26 - Prob. 26.27PCh. 26 - Prob. 26.28PCh. 26 - Prob. 26.29PCh. 26 - Prob. 26.30PCh. 26 - Prob. 26.31PCh. 26 - Prob. 26.32PCh. 26 - Prob. 26.33P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give numerical answers to the following questions about the structure of a cholesterol molecule. a. How many six-membered rings arc present? b. How many amide linkages are present? c. How many hydroxyl substituents are present? d. How many total functional groups are present?arrow_forwardDraw the structure of each of the following compounds. an α-N-glycoside formed from D-arabinose and C6H5CH2NH2arrow_forwardIn an aqueous solution, d-glucose exists in equilibrium with two six-membered ring compounds. Draw the structures of these compounds.arrow_forward
- Between fatty acid A and B, Which is most likely solid at room temperature? CH3(CH2)18CO2H CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7CO2Harrow_forwardWrite the structure of the following compounds: γ-lactolarrow_forwardArachidonic acid is a 20-carbon polyunsaturated essential fatty acid with 4 double bonds at positions omega-6, omega-9, omega-12, and omega-15. Draw the naturally occurring form of this fat.arrow_forward
- Draw and label a strcture of: Cervonic acid 22:6 Δ4,7,10,13,16,19. This is a fatty acid found in cold fish.arrow_forwardCan different fatty acids form esters on the same glycerol molecule? Is it possible for a trans-fatty acid to be synthesized from a cis-fatty acid? if so how? How many hydroxyl groups does the Sphingomyelin backbone contain?arrow_forward1. Which of the following are saponifiable lipids? a) triacylglycerols; f) stepoids3; b) prostaglandins3B g) glycolipids; c) waxes;B h) terpenes; d) leukotrienes; i) phosphoglycerides; e) sphyngolipids; k) plasmalogens?arrow_forward
- Draw the skeletal structures of the hydrolysis products carboxylic acid and alcohol formed when the wax is treated with aqueous sulfuric acid. CH3(CH2)12COO(CH2)25CH3arrow_forward4. The ester you drew in Question #1 has a melting point range of 32-35 °C. The methyl ester of mono-unsaturated palmitoleic acid (IUPAC name: hexadec-9enoic acid) has a considerably lower melting point range: -0.5-+0.5 °C. a) Explain what it means for a fatty acid to the "mono-unsaturated." b) Illustrate your explanation by drawing the structure of the methyl ester of octadec-11-enoic acid. (Be sure to draw the geometric stereoisomer that is commonly found in nature.)arrow_forward19-22 What is the relationship between degree of unsaturation and melting point for fatty acids? te associat locule?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,