
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 26, Problem 26.24P
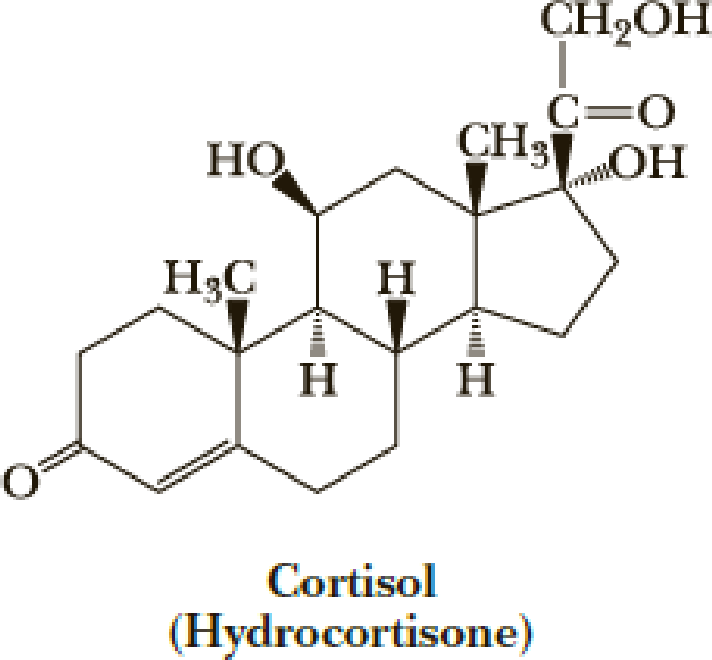
Following is a structural formula for cortisol (hydrocortisone). Draw a stereo-representation of this molecule showing the conformations of the five- and six-membered rings.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Organic Chemistry HW:
CANNOT BE HAND DRAWN
2,6-dimethyloct-2-ene
Provide a detailed typed explanation of
Stereoisomers show the expanded structure of your molecule.
Calculate the maximum number of possible stereoisomers of your molecule using the following formula:
Maximum number of possible stereoisomers = 2n (where n= the number of chiral carbons in your molecule). This calculation does not include E- or Z- isomers for any compounds containing double bonds
Type or using a computer program "draw" the possible stereoisomers of the molecule.
Note that E-, Z- isomers of each stereoisomer are also possible and would not be accounted for by the formula above; draw any E- or Z- isomers.
Draw molecules for the formula C5H10O with functional group alcohol and an ether with an R chiral center.
Name the isomers of the compound with the closed formula C4H9Cl. Which of these isomers can have a chiral structure? Examine it from a stereochemical point of view.
Chapter 26 Solutions
Organic Chemistry
Ch. 26.1 - Prob. 26.1PCh. 26.6 - Prob. AQCh. 26.6 - Prob. BQCh. 26.6 - Prob. CQCh. 26.6 - Prob. DQCh. 26.6 - Prob. FQCh. 26 - Prob. 26.2PCh. 26 - Identify the hydrophobic and hydrophilic region(s)...Ch. 26 - Prob. 26.4PCh. 26 - Prob. 26.5P
Ch. 26 - Prob. 26.6PCh. 26 - Prob. 26.7PCh. 26 - Prob. 26.8PCh. 26 - Prob. 26.9PCh. 26 - How many moles of H2 are used in the catalytic...Ch. 26 - Prob. 26.11PCh. 26 - Prob. 26.12PCh. 26 - Prob. 26.13PCh. 26 - Prob. 26.14PCh. 26 - Prob. 26.15PCh. 26 - Prob. 26.16PCh. 26 - Prob. 26.17PCh. 26 - Prob. 26.18PCh. 26 - Prob. 26.20PCh. 26 - Prob. 26.21PCh. 26 - Prob. 26.22PCh. 26 - Prob. 26.23PCh. 26 - Following is a structural formula for cortisol...Ch. 26 - Prob. 26.25PCh. 26 - Draw the structural formula of a lecithin...Ch. 26 - Prob. 26.27PCh. 26 - Prob. 26.28PCh. 26 - Prob. 26.29PCh. 26 - Prob. 26.30PCh. 26 - Prob. 26.31PCh. 26 - Prob. 26.32PCh. 26 - Prob. 26.33P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- There are 17 possible alkene isomers with the formula C6H12. Draw structures of the five isomers in which the longest chain has six carbon atoms, and give the name of each. Are any of these isomers chiral? (There are also eight isomers in which the longest chain has five carbon atoms, and four isomers in which the longest chain has four carbon atoms. How many can you find?)arrow_forwardCompound: 1-(isopropyl)-2-methylcyclohexane Sketch the line drawing of all the isomers of this compound (showing the 3D nature of the compound with wedges and dashes)arrow_forwardWrite a conformational structure for 1,2,3-trimethylcyclohexane in which all the methyl groups are axial and then show its more stable conformation.arrow_forward
- A student drew a noncyclic compound that has the molecular formula C7H14.This molecular formula is representative of an alkane, alkene, alkyne or aromatic and would be classified as saturated or unsaturated ?arrow_forwardExplain the concept of Cyclohexane Conformations ?arrow_forwardHow To Draw the Two Conformations for a Substituted Cyclohexane ?arrow_forward
- There are 11 structures (ignoring stereoisomerism) with the formula C4H8O that have no carbon branches. Draw the structures and identify the functional groups in each.arrow_forwardDraw and name the six isomeric cyclopentane of molecular formula C7H14. These will include four constitutional isomers, of which two show geometric (cis-trans) stereoisomerism.arrow_forwardDo conformational isomers count as stereoisomers? For example, if we consider disubstituted cyclohexane and not care about chirality, does it yield three conformational isomers, axial-axial, axial-equatorial and equatorial-equatorial?arrow_forward
- Give the systematic name for the compound 2.2 ignoring any stereochemical designationarrow_forwardDraw structural formulas for the cis and trans isomers of hydrindane. Show each ring in its most stable conformation. Which of these isomers is more stable?arrow_forwardHow many degrees of unsaturation does C8H10 have?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License