
What starting materials are needed to synthesize each compound by a thermal
a.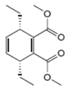 b.
b. 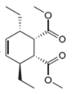 c.
c. 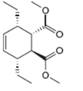
(a)
Interpretation: The starting materials that are needed to synthesize the given compound by a thermal
Concept introduction: A chemical reaction that involves
Answer to Problem 27.41P
The starting materials that are needed to synthesize the given compound by a thermal
Explanation of Solution
The given product is shown below.
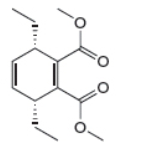
Figure 1
Retro synthesis can be carried out to identify the diene and dienophile for the given product.
The reaction that shows the disconnection approach of the given product is shown below.
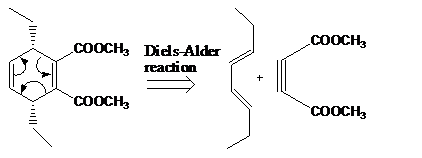
Figure 2
In the given product, the ring is opened due to the rearrangement of
The starting materials that are needed to synthesize the given compound by a thermal
(b)
Interpretation: The starting materials that are needed to synthesize the given compound by a thermal
Concept introduction: A chemical reaction that involves
Answer to Problem 27.41P
The starting materials that are needed to synthesize the given compound by a thermal
Explanation of Solution
The given product is shown below.
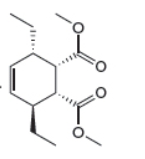
Figure 3
Retro synthesis can be carried out to identify the diene and dienophile for the given product.
The reaction that shows the disconnection approach of the given product is shown below.
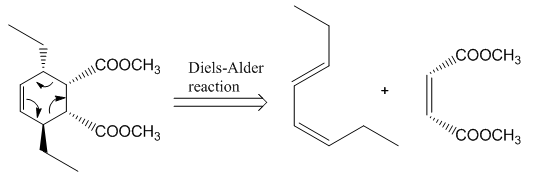
Figure 4
In the given product, the ring is opened due to the rearrangement of
Thus, the starting materials that are needed to synthesize the given compound by a thermal
The starting materials that are needed to synthesize the given compound by a thermal
(c)
Interpretation: The starting materials that are needed to synthesize the given compound by a thermal
Concept introduction: A chemical reaction that involves
Answer to Problem 27.41P
The starting materials that are needed to synthesize the given compound by a thermal
Explanation of Solution
The given product is shown below.
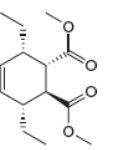
Figure 5
Retro synthesis can be carried out to identify the diene and dienophile for the given product.
The reaction that shows the disconnection approach of the given product is shown below.
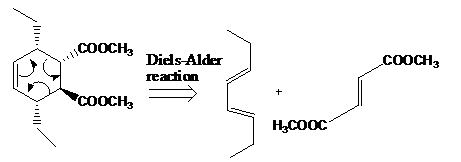
Figure 6
In the given product, the ring is opened due to the rearrangement of
Thus, the starting materials that are needed to synthesize the given compound by a thermal
The starting materials that are needed to synthesize the given compound by a thermal
Want to see more full solutions like this?
Chapter 27 Solutions
ORGANIC CHEMISTRY >ACCESS CODE<
- (a) What alkene yields A and B when it is treated with Br2 in CCl4? (b) What alkene yields C and D under the same conditions?arrow_forwardFor alkenes A, B, C, and D: (a) Rank A—D in order of increasing heat ofhydrogenation; (b) rank A—D in order of increasing rate of reaction withH2, Pd-C; (c) draw the products formed when each alkene is treated withozone, followed by Zn, H2O.arrow_forwardDraw a stepwise mechanism for the conversion of dihalide A andcyclohexane-1,4-diol to polyether B in the presence of AlCl3arrow_forward
- Hydrogenation of alkene A with D2 in the presence of Pd-C affords a single product B. Keeping this result in mind, what compound is formed whenA is treated with each reagent: (a) mCPBA; (b) Br2, H2O followed by base? Explain these results.arrow_forwardWhat product is formed when each compound undergoes thermal electrocyclic ring opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.arrow_forwardDevise a stepwise synthesis of attached compound from dicyclopentadieneusing a Diels–Alder reaction as one step. You may also use organiccompounds having ≤ 4 C's, and any required organic or inorganicreagents.arrow_forward
- Suzuki coupling of aryl iodide A and vinylborane B affords compound C, which is converted to D in the presence of aqueous acid. Identify compounds C and D and draw a stepwise mechanism for the conversion of C to D.arrow_forwardDraw the product (including stereochemistry) formed from each pair of reactants in a thermal [4 + 2] cycloaddition reaction.arrow_forwarda. How many stereoisomers are formed from the reaction of cyclohexene with NBS?b. How many stereoisomers are formed from the reaction of 3-methylcyclohexene with NBS?arrow_forward
- What alkene yields A and B when it is treated with Br2 in CCl4?arrow_forward(a) Draw all constitutional isomers formed by monochlorination of each alkane with Cl2 and hν. (b) Draw the major monobromination product formed by heating each alkane with Br2.arrow_forwardFor alkenes A, B, C, and D: (a) Rank A—D in order of increasing heat of hydrogenation; (b) rank A—D in order of increasing rate of reaction with H2, Pd-C; (c) draw the products formed when each alkene is treated with ozone, followed by Zn, H2O.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
