
Concept explainers
What is the empirical formula of the compound shown?
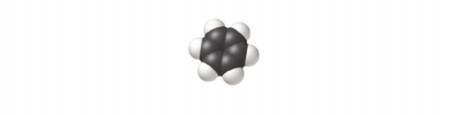
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
ALEKS 360-ACCESS (52 WEEKS)
- A compound has a molar mass of 155.5 g/mol. The empirical formula of the compound is C₂H₇. How many carbon atoms are in the true molecular formula of the compound?arrow_forwardWhat is the empirical formula for a sample containing 79.842% carbon and 19.943% hydrogen? a.) CH3 b.) C3H8 c.) C2H6 d.) CH e.) CH4arrow_forwardWhat is the mass percent C in isooctane, C(CH₃)₃CH₂CH(CH₃)₂. a. 1.588 % C b. 8.412 % C c. 84.12 % C d. 15.88 % C e. 74.04 % Carrow_forward
- What is the empirical formula of a mineral contg. 3.37% H2O; 19.19% Al2O3; 21.00% CaO; and 56.53% SiO3?arrow_forwardThe molecular formula and the empirical formula for this hydrocarbon are a. C5 H10 and CH2 b. C5 H10 and CH3 c. C4H8 and CH2 d. C4H8 and CH3arrow_forward_____ 3. What is the empirical formula for the compound with the following molecular formula? C6H18O3 C6H18O3 (C2H6C)3 C2H6O CHOarrow_forward
- Calculate the molecular formula of a compound that has an empirical formula of P2O5arrow_forward8.How many carbon atoms in 70 molecules of carbonic acid, H2CO3 are. A) 100 B) 4.8x1025 C) 70 D) 25 E) 6.02 X1023arrow_forwardDetermine the molecular formula of a compound that is 49.48% carbon, 5.19% hydrogen, 28.85% nitrogen, and 16.48% oxygen. The molecular weight is 194.19 g/mol. C8H12N4O2 C4H5N2O C8H10N2O2 C8H10N4O2arrow_forward
- Find the molecular formula of the following compounds 9. A class of compounds called sodium metaphosphates were used as additives to detergents to improve cleaning ability. One of them has a molecular mass of 612g. Analysis shows the composition to be 22.5% Na, 30.4% P, and 47.1% O. Determine the molecular formula of this compound. (answer please and please if you're going to write it make sure that I can read it)arrow_forwardA compound is 57.14% C, 6.16% H, 9.52% N, and 27.18% O. The molar mass is 588.6 g/mol. What is the molecular formula? Group of answer choices C14H18N5O2 C28H36N4O10 C7H9NO3 C14H18N2O5 C7H9NO5arrow_forwardDetermine the empirical formula for the following compound: C8H10N4O2 A. C8H10N8O4 B. C2H5NO C. CHNO D. C4H5N4O2 E. C8H10N4O2arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
