
Concept explainers
a)
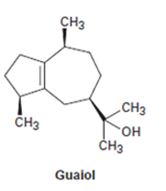
Interpretation:
At what positions the terpenoid guaiol will be labeled if isopentyl diphosphate and dimethylallyl diphosphate from which they are synthesized are isotopically labeled at the diphosphate bearing carbon atom (C1).
Concept introduction:
In the formation of terpenes every fourth carbon of one dimethylallyl phosphate (formed by the isomerization of isopentyl diphosphate) combines with the first carbon of the isopentyl phosphate molecule. Hence if the diphosphate bearing carbon is labeled, every fourth carbon in a terpene will be a labeled carbon.
To give:
The positions at which the terpenoid guaiol will be labeled if isopentyl diphosphate and dimethylallyl diphosphate from which it is synthesized are isotopically labeled at the diphosphate bearing carbon atom (C1).
b)
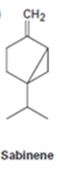
Interpretation:
At what positions the terpenoid sabinene will be labeled if isopentyl diphosphate and dimethylallyl diphosphate from which they are synthesized are isotopically labeled at the diphosphate bearing carbon atom (C1).
Concept introduction:
In the formation of terpenes every fourth carbon of one dimethylallyl phosphate (formed by the isomerization of isopentyl diphosphate) combines with the first carbon of the isopentyl phosphate molecule. Hence if the diphosphate bearing carbon is labeled, every fourth carbon in a terpene will be a labeled carbon.
To give:
The positions at which the terpenoid sabinene will be labeled if isopentyl diphosphate and dimethylallyl diphosphate from which it is synthesized are isotopically labeled at the diphosphate bearing carbon atom (C1).
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- 17-74 Glucose, C6H12O6, contains an aldehyde group but exists predominantly in the form of the cyclic hemiacetal shown here. We will discuss this cyclic form of glucose in Chapter 20. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. (a) Which carbon in glucose provides the —OH group and which provides the —CHO group? (b) Draw the alternative chair confirmations of D-glucose and state which of the two is the more stable.arrow_forwardRing-A is considered a deoxy-pyranoside because it is missing an alcohol functional group on which carbon of ring-A? Use standard carbohydrate numbering. E в он он Oleandrin is a toxic cardiac glycoside found in the poisonous plant, oleander (Nerium oleander L). It has a very long IUPAC name: acetic acid [(35,5R, 10S,13R,14S,16S,17'R)-14-hydroxy-3-[[(2R,4S,5S,6S)-5-hydroxy-4-methoxy-6-methyl- 2-tetrahydropyranyl]oxy]-10,13-dimethyl-17-(5-oxo-2H-furan-3-y)-1,2,3,4,5,6,7,8,9,11,12,15,16,17- tetradecahydrocyclopenta[a]phenanthren-16-yl] ester B.arrow_forwardCh19-5: Which of the compounds is the intermediate called hemiacetal of the following reaction? 요 HO OH H₂SO (-H₂O) OH HO OH HO HOO O OH OHarrow_forward
- Trimyristin is obtained from coconut oil and has the molecular formula C45H86O6. On being heated with aqueous sodium hydroxide followed by acidification, trimyristin was converted to glycerol and tetradecanoic acid as the only products. What is the structure of trimyristin?arrow_forward13-18.) Given the structures of the naturally occurring aldoses, list at least three examples of the following: a) diastereomers b) epimers Three carbons Four carbons Five carbons H--OH H--OH HO--H HO--H но-н H--OH ČH,OH H-C-OH HO--H H--OH H--OH ČH,OH -Erythrose D-Threose HO--H H-C-OH H-C-OH H--OH H--OH ČH,OH H--OH H-C-OH ČH,OH CH,OH CH,OH ČH,OH »Glyceraldehyde D-Ribose D-Arabinose D-Xylose D-Lyxose Six carbons H-C-OH HO-C-H H-C-OH HO--H HO-C-H H-C-OH HO-C-H H--OH HO-C-H H-C-OH HO-C-H HO-H HO--H HO--H HO--H H--OH CH,OH H-OH HO-H H-OH H--OH H--OH H-C-OH H-OH CH,OH H--OH CH,OH H-C-OH H OH CH,OH H-C-OH H--OH CH,OH HO-C-H H--OH CH,OH H-C-OH H--OH CH,OH D-Galactose -Talose CH,OH D-Allose D-Altrose D-Glucose D-Mannose D-Gulose D-Idose D-Aldoses (a)arrow_forwardIf a carboxylic acid is dissolved in isotopically labeled methanol (CH3 18OH) and an acid catalyst is added, where will the label reside in the product?arrow_forward
- Which of the following functional group (FG) will produce a ketone upon oxidation with K2CrO7? (a) RX (b) 10OH (b) 20OH (c) 30OHarrow_forward17-14 Following are structural formulas for two steroid hormones. (a) Name the functional groups in each. (b) Mark all stereocenters in each hormone and state how many stereoisomers are possible for each.arrow_forward13-29 Show that if you add Steps 2a and 2b of the radical- chain mechanism for the autoxidation of a fatty acid hydrocarbon chain, you arrive at the following net equation: H I —CH2CH=CH—CH— + 0—0 • » Section of a fatty acid Oxygen hydrocarbon chain O—O—H I —CH2CH=CH—CH— A hydroperoxidearrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


