
(a)
Interpretation: The structure of the polymer formed by chain-growth
Concept introduction: The simpler units which combine to form
Answer to Problem 24P
The structure of the polymer formed by chain-growth polymerization of given monomer is,
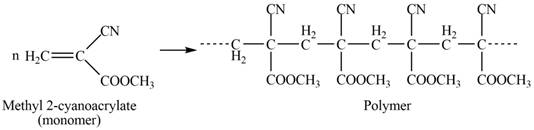
Explanation of Solution
The given molecule is
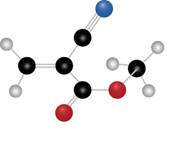
Figure 1
The blue coloured ball has three bonds. So, this is the nitrogen atom. The red coloured ball has two bonds. So, this is the oxygen atom. The black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure of the compound is,
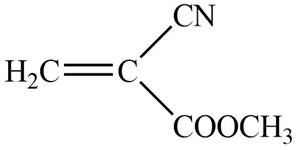
Figure 2
Chain-growth polymerization is formed by the repetitive addition of monomer units containing double or triple bonds. If the monomer units undergoing addition are the same, the polymer is known as
The polymer formed by chain-growth polymerization of given monomer is shown below.
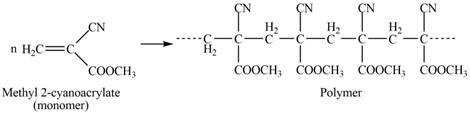
Figure 3
The structure of the polymer formed by chain-growth polymerization of given monomer is shown in Figure 3.
(b)
Interpretation: The structure of the polymer formed by chain-growth polymerization of given monomer is to be drawn.
Concept introduction: The simpler units which combine to form polymers are known as monomers. The process by which respective monomers combine to form polymers is known as polymerization.
Answer to Problem 24P
The structure of the polymer formed by chain-growth polymerization of given monomer is,
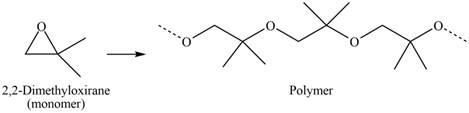
Explanation of Solution
The given molecule is
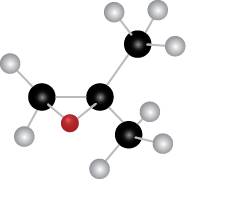
Figure 4
The red coloured ball has two bonds. So, this is the oxygen atom. The black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure of the compound is,
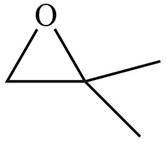
Figure 5
Chain-growth polymerization is formed by the repetitive addition of monomer units containing double or triple bonds. If the monomer units undergoing addition are the same, the polymer is known as homopolymer.
The polymer formed by chain-growth polymerization of given monomer is shown below.
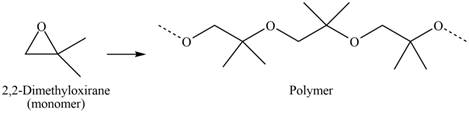
Figure 6
The structure of the polymer formed by chain-growth polymerization of given monomer is shown in Figure 6.
Want to see more full solutions like this?
Chapter 28 Solutions
LL ORG CHEM
- Super glue contains methyl cyanoacrylate, which readily polymerizes upon exposure to traces of water or alcohols on the surfaces to be bonded together. The polymer provides a strong bond between the two surfaces. Draw the structure of the polymer formed by methyl cyanoacrylate.arrow_forwardDraw the structure of the polymer formed by step-growth polymerization of each monomer or pair of monomers. а. b.arrow_forwardWhat monomer is needed to form each polymer?arrow_forward
- A particularly strong and rigid polyester used for electronic parts is marketed under the trade name Glyptal. It is a polymer of terephthalic acid and glycerol. Draw a segment of the polymer and explain why it is so strong.arrow_forward7. During condensation polymerization, many monomers join together. What is lost when two mon omers join together via a condensation reaction? carbon dioxide b. hydrochloric acid c. hydrogen gas а. d. waterarrow_forwardWhat polymer is formed by chain-growth polymerization of each monomer?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





