
Concept explainers
Interpretation:
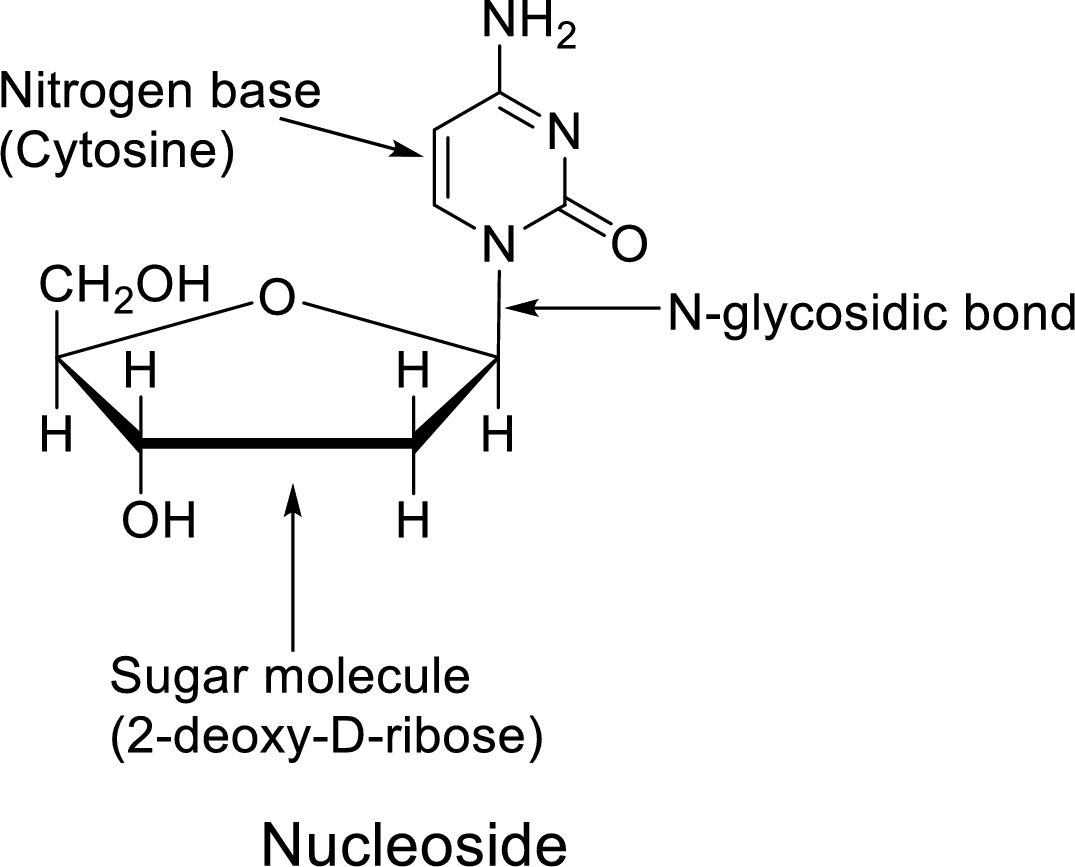
In dilute acid, the glycosidic bond of Nucleoside undergoes hydrolysis to give a pentose and a heterocyclic
Concept Introduction:
The Nucleoside is the component of the
Nucleoside = Nitrogen base + Sugar molecule
Trending nowThis is a popular solution!

Chapter 28 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- 22-97 Gelatin is derived from collagen by denaturation. Is a gelatin dessert likely to be a good source of dietary protein?arrow_forwardWhy can isomaltose be easily hydrolyzed through acid hydrolysis while trehalose cannot undergo acid hydrolysis?arrow_forwardThe hydrolysis of an ester can be sped up by both acidic and basic conditions. Aminolysis of an ester can be sped up by acidic conditions, but not by basic conditions. Explain why.arrow_forward
- An octapeptide contains the following amino acids: Arg, Glu, His, Ile, Leu, Phe, Tyr, and Val. Carboxypeptidase treatment of the octapeptide forms Phe and a heptapeptide. Treatment of the octapeptide with chymotrypsin forms two tetrapeptides, A and B. Treatment of A with trypsin yields two dipeptides, C and D. Edman degradation cleaves the following amino acids from each peptide: Glu (octapeptide), Glu (A), Ile (B), Glu (C), and Val (D). Partial hydrolysis of tetrapeptide B forms Ile–Leu in addition to other products. Deduce the structure of the octapeptide and fragments A–D.arrow_forwardContrast the structure of glycogen and chitin. Drag the appropriate items to their respective rows. Glycogen Chitin Monomers connect by B-1,4-glycosidic linkages Monomers connect by alpha-1,4-glycosidic linkages Polymers interact with one another via hydrogen bonds Consists of modified glucose residues (NAG) Contains branches arising from alpha-1,6-glycosidic linkages Consists of glucose residuesarrow_forward22-61 Polyglutamic acid (a polypeptide chain made only of glutamic acid residues) has an a-helix conformation below pH 6.0 and a random-coil conformation above pH 6.0. What is the reason for this conformational change?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



