
Concept explainers
(a)
Interpretation: The products formed by the treatment of D-altrose with
Concept introduction: The
Answer to Problem 28.51P
The products formed by the treatment of D-altrose with
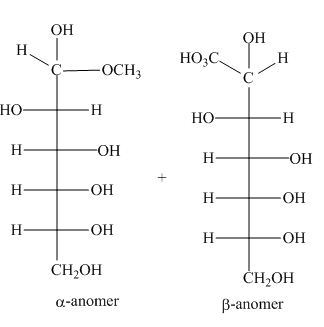
Explanation of Solution
The
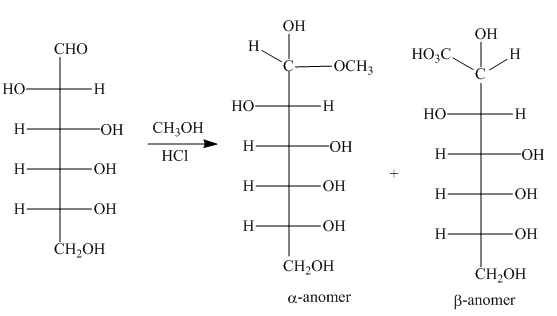
Figure 1
The products formed by the treatment of D-altrose with
(b)
Interpretation: The products formed by the treatment of D-altrose with
Concept introduction: The
Answer to Problem 28.51P
The products formed by the treatment of D-altrose with
Explanation of Solution
The
The products formed by the treatment of D- altrose with
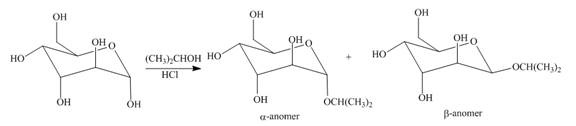
Figure 2
The products formed by the treatment of D-altrose with
(c)
Interpretation: The products formed by the treatment of D-altrose with
Concept introduction: The substitution reaction involves the replacement of one functional group by other functional group. In nucleophilic substitution an electron rich species attack the species that is deficient in electrons. The electrophile and the leaving group together form a substrate. The nucleophile attacks over the substrate and there occurs the removal of leaving group from the substrate.
Answer to Problem 28.51P
The product formed by the treatment of D-altrose with
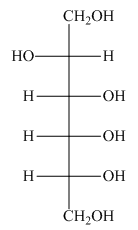
Explanation of Solution
The product formed by the treatment of D-altrose with
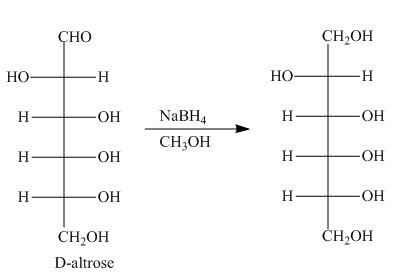
Figure 3
In the given reaction, sodium borohydride is used as a reducing agent. Sodium borohydride is used to for the reduction of carbonyl compounds to alcohols.
The product formed by the treatment of D-altrose with
(d)
Interpretation: The products formed by the treatment of D-altrose with
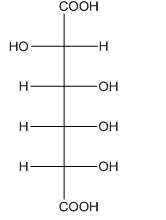
Concept introduction: The aldehyde group of aldoses oxidizes to carboxyl group on treatment with
Answer to Problem 28.51P
The product formed by the treatment of D-altrose with
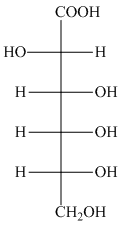
Explanation of Solution
The oxidation of aldehyde group of aldoses leads to the formation of aldonic acid, in which the terminal carbon atoms are substituted by carboxyl group and alcoholic group. The product formed by the treatment of D-altrose with
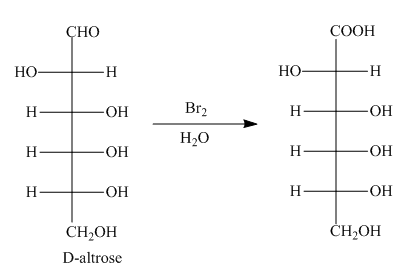
Figure 4
The product formed by the treatment of D-altrose with
(e)
Interpretation: The products formed by the treatment of D-altrose with
Concept introduction: The aldehyde group and primary alcohol of aldoses oxidizes to carboxyl groups on treatment with warm
Answer to Problem 28.51P
The product formed by the treatment of D-altrose with
Explanation of Solution
The oxidation of aldehyde group and primary alcohol of aldoses leads to the formation of aldaric acid, in which the terminal carbon atoms are substituted by carboxyl groups. The product formed by the treatment of D-altrose with
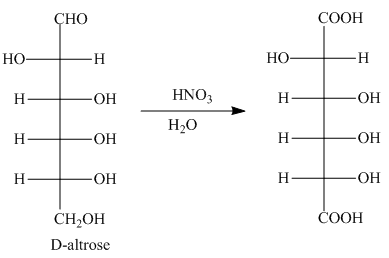
Figure 5
The product formed by the treatment of D-altrose with
(f)
Interpretation: The products formed by the treatment of D-altrose with [1]
Concept introduction: The
Answer to Problem 28.51P
The product formed by the treatment of D-altrose with [1]
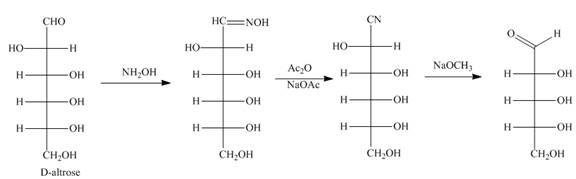
Explanation of Solution
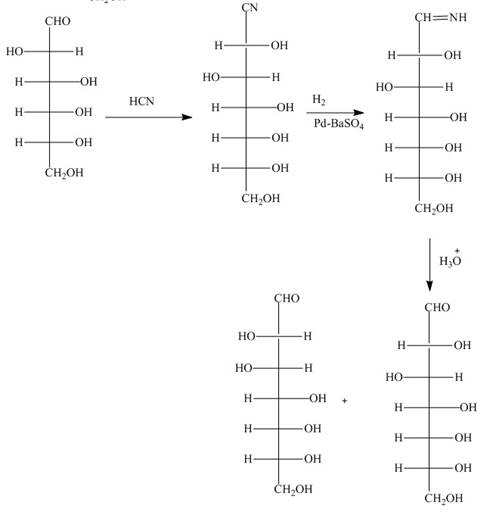
The reaction of D-altrose with hydroxylamine results in the formation of oxime. The second step is the dehydration of oxime to nitriles. The reaction of nitrile with
The product formed by the treatment of D-altrose with [1]
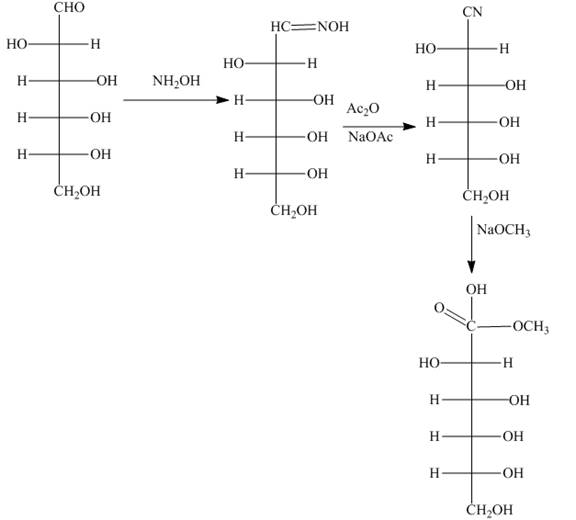
Figure 6
The product formed by the treatment of D-altrose with [1]
(g)
Interpretation: The products formed by the treatment of D-altrose with [1]
Concept introduction: The
Answer to Problem 28.51P
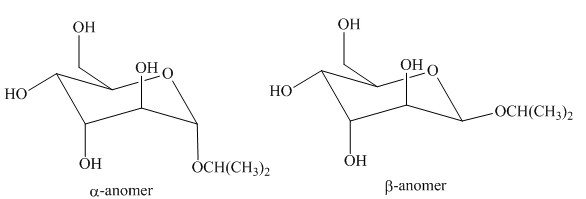
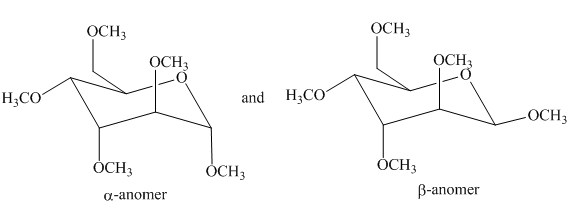
The products formed by the treatment of D-altrose with [1]
Explanation of Solution
The products formed by the treatment of D-altrose with [1]
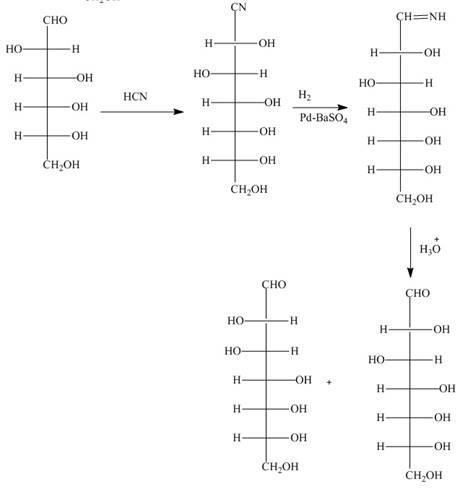
Figure 7
The products formed by the treatment of D-altrose with [1]
(h)
Interpretation: The products formed by the treatment of D-altrose with
Concept introduction: The
Answer to Problem 28.51P
The products formed by the treatment of D-altrose with
Explanation of Solution
The
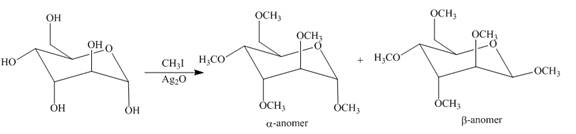
Figure 8
The products formed by the treatment of D-altrose with
(i)
Interpretation: The products formed by the treatment of D-altrose with
Concept introduction: The
Answer to Problem 28.51P
The products formed by the treatment of D-altrose with
Explanation of Solution
The
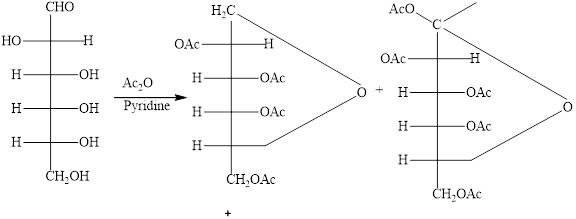
Figure 9
The products formed by the treatment of D-altrose with
(j)
Interpretation: The products formed by the treatment of D-altrose with
Concept introduction: The
Answer to Problem 28.51P
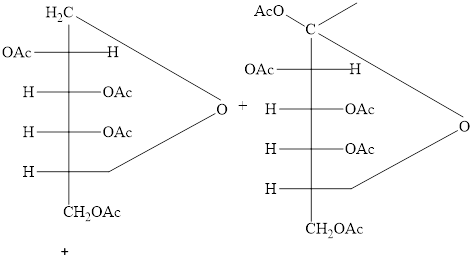
The products formed by the treatment of D-altrose with
Explanation of Solution
The
Figure 10
The products formed by the treatment of D-altrose with
Want to see more full solutions like this?
Chapter 28 Solutions
Organic Chemistry
- Draw the product formed when (CH3)2CHOH is treated with each reagent (a, b and c)arrow_forwardDraw the product formed when (CH3)2CHOH is treated with each reagent (d, e and f)arrow_forwardDraw the products formed when α-D-gulose is treated with each reagent. a. CH3I, Ag2O b. CH3OH, HCl c. Ac2O, pyridine d. The product in (a), then H3O+ e. The product in (b), then Ac2O, pyridine f. The product in (d), then C6H5CH2Cl, Ag2Oarrow_forward
- Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH c.HCl + ZnCl2 d.HBr e.SOCl2, pyridine f.PBr3 g.TsCl, pyridine h. [1] NaH; [2] CH3CH2Br [1] i.TsCl, pyridine; [2] NaSH j.POCl3, pyridinearrow_forwardDraw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.arrow_forwardDraw the product formed when (CH3)2CHOH is treated with following reagent. POCl3, pyridinearrow_forward
