
Concept explainers
(a)
Interpretation:
The higher energy should be identified.
Concept introduction:
The shorter the wavelength, the higher the energy, the longer the wavelength, the lower the energy.
Wave length region is shown in figure 1.
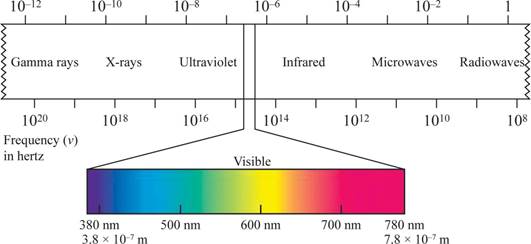
Figure 1
(b)
Interpretation:
The higher energy should be identified.
Concept introduction:
The shorter the wavelength, the higher the energy, the longer the wavelength, the lower the energy.
Wave length region is shown in figure 1.
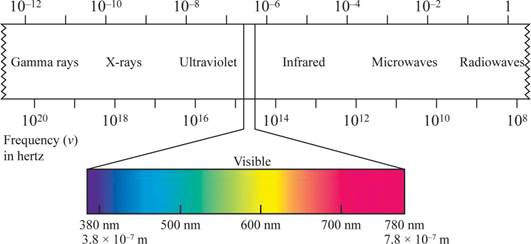
Figure 1
(c)
Interpretation:
The higher energy should be identified.
Concept introduction:
The shorter the wavelength, the higher the energy, the longer the wavelength, the lower the energy.
Wave length region is shown in figure 1.
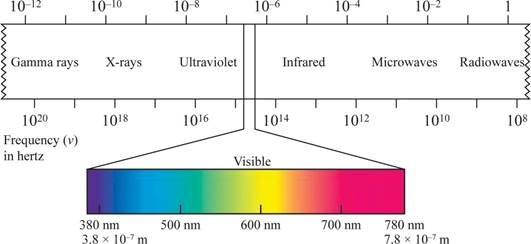
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- How many half-lives would have to elapse for a sample of a radioactive isotope to decrease from an activity of 224 cpm to an activity of 7 cpm?arrow_forwardIn your own words define what is Dark matter?arrow_forwardWhat type of energy is used by your body when you eat food? Group of answer choices chemical energy thermal energy gravitational energy electrical energy What type of energy comes from the motion of tiny particles of matter? Group of answer choices thermal energy Sound energy gravitational energy nuclear energyarrow_forward
- Which of the following statements is true? 1) The shortest wavelength of light is the ultraviolet ray, which is the highest in energy. 2) The longest wavelength of light is the ultraviolet ray, which is the highest in energy 3) The shortest wavelength of light is the visible light, which is the highest in energy 4 ) The shortest wavelength of light is the infrared ray, which is the highest in energy. 5) The longest wavelength of light is the infrared raywhich is the highest in energy .arrow_forwardAlbert Einstein’s equation for the conservation of matter and energy (E = mc2) predicts that if the mass of uranium to be split in a nuclear fission reaction were to double, then: the amount of energy released would be decreased to one-fourth the amount of energy released would be decreased to one-half the amount of energy released would be increased four-fold the amount of energy released would be increased two-fold the amount of energy released would remain the samearrow_forwardMolecules that absorb electromagnetic energy have structural components called _____________.arrow_forward
- The earth is moving at approximately what speed in miles per hour arrow_forwardA slice of pizza has 500 kcal. If we could burn the pizza and use all the heat to warm a 50-L container of cold water, what would be the approximate increase in the temperature of the water? (note; A Liter of cold water weighs about 1 kg) a. 500 C b. 50C c. 10C d.100Carrow_forwardPolonium is a rare element with 33 radioisotopes. The most common one, 210Po, has 82 protons and 128 neutrons. When 210Po decays, it emits an alpha particle, which is a helium nucleus (2 protons and 2 neutrons). 210Po decay is tricky to detect because alpha particles do not carry very much energy compared to other forms of radiation. For example, they can be stopped by a single sheet of paper or a few inches of air. That is one reason that authorities failed to discover toxic amounts of 210Po in the body of former KGB agent Alexander Litvinenko until after he died suddenly and mysteriously in 2006. What element does an atom of 210Po become after it emits an alpha particle?arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax




