
Concept explainers
Decide whether each of the following is water-soluble. If soluble, tell what ions are produced when the compound dissolves in water.
(a) NiCl2
(b) Cr(NO3)3
(c) Pb(NO3)2
(d) BaSO4
(a)
Interpretation:
Water solubility of the given compounds and ions produced by the compounds should be analyzed.
Concept introduction:
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 16PS
Water soluble compound. Produced ions are
Explanation of Solution
The given compound Nickel (II) chloride which is soluble in water.
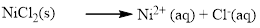
(b)
Interpretation:
Water solubility of the given compounds and ions produced by the compounds should be analyzed.
Concept introduction:
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 16PS
Water soluble compound. Produced ions are
Explanation of Solution
The given compound is Chromium (III) nitrate which is soluble in water.
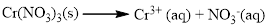
(c)
Interpretation:
Water solubility of the given compounds and ions produced by the compounds should be analyzed.
Concept introduction:
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 16PS
Water soluble compound. Produced ions are.
Explanation of Solution
The given compound is Lead (II) nitrate which is soluble in water.
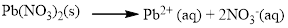
(d)
Interpretation:
Water solubility of the given compounds and ions produced by the compounds should be analyzed.
Concept introduction:
Most of the ionic compounds are soluble in water, very few of the ionic compounds are sparingly soluble, and some of the ionic compounds are insoluble in water. When it is soluble in water ions gets separated in the solution.
Soluble compounds in water
Almost all the salts of
Almost all the salts of
Salts of F- are soluble. But some of the fluoride salt of
Salts of
Insoluble compounds in water:
Most of the salts of
Most of the metal hydroxides and oxides are insoluble in water bit some of the alkali metal hydroxides,
Answer to Problem 16PS
This compound insoluble in water.
Explanation of Solution
The given compound is Barium sulfate which is insoluble in water.
Want to see more full solutions like this?
Chapter 3 Solutions
EP CHEMISTRY+CHEMICAL REACTIVITY-OWLV2
- Will a precipitate form when solutions of Ba(NO3)2 and KOH are mixed?arrow_forwardWill a precipitate form if solutions of the soluble saltsPb(NO3)2 and KI are mixed?arrow_forwardThe hardness of water (hardness count) is usually expressed in parts per million (by mass) of CaCO3, which is equivalent to milligrams of CaCO3 per liter of water. What is the molar concentration of Ca2+ ions in a water samplewith a hardness count of 175 mg CaCO3/L?arrow_forward
- When a piece of silver is submerged in a solution of lead (II) nitrate (Pb(NO3)2), will it become coated in lead?arrow_forwardThe US Environmental Protection Agency (EPA) places limits on the quantities of toxic substances that may be discharged into the sewer system. Limits have been established for a variety of substances, including hexavalent chromium, which is limited to 0.50 mg/L. If an industry is discharging hexavalent chromium as potassium dichromate (K2Cr2O7), what is the maximum permissible molarity of that substance?arrow_forwardLead compounds used to be use of paint pigments (lead based paints), but sre now prohibited in homes because the Pb (II) ion is toxic. Which of the followint compounds would yeild tbe greast amount of Pb (II) ions when added to tne same quantity of water (assuming some undissolved solid always remains) ? A) PbCrO4 B) PbSO4 C) PbSarrow_forward
- When aqueous ammonium carbonate, (NH4)2CO3, is treated with potassium hydroxide, KOH, the products are potassium carbonate, K2CO3, water and ammonia. What is the net ionic equation for this reaction?arrow_forwardwhat chemical could you add and what reactions would occur that would allow you to distinguish between unlabeled bottles of solutions of kno3 and zn(no3)2arrow_forwardIs Fe(No2) a strong, weak, or nonelectrolyte compound upon dissolving in water? What about NH3(aq)?arrow_forward
- Excess Na2SO4(aq) is added to a 2.43×102 mL sample of industrial waste containing Ba2+ ions. If 20.5 g of BaSO4(s) are precipitated from the reaction, what was the molar concentration of Ba2+ in the original sample?arrow_forwardThe hardness of water (hardness count) is usually expressed in parts per million (by mass) of CaCO3, which is equivalent to milligrams of CaCO3 per liter of water. What is the concentration of Ca2+ ions in water sample with a hardness count of 188 mg CaCO3/L?arrow_forwardA 0,4500 g sample of a pesticide (DDT, C14H9Cl5, MM 354,49 g mol^-1) was decomposed with metallic sodium in alcohol and the released chloride ions were precipitated as AgCl (MM 143,32 g mol-1) . If the precipitate mass corresponds to 0,1706 g of AgCl, calculate the percentage of DDT in the sample.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
