
Concept explainers
Refer to the color scheme given Figure 3-3, and give the molecular formulas for molecules whose ball-and-stick models are given here.
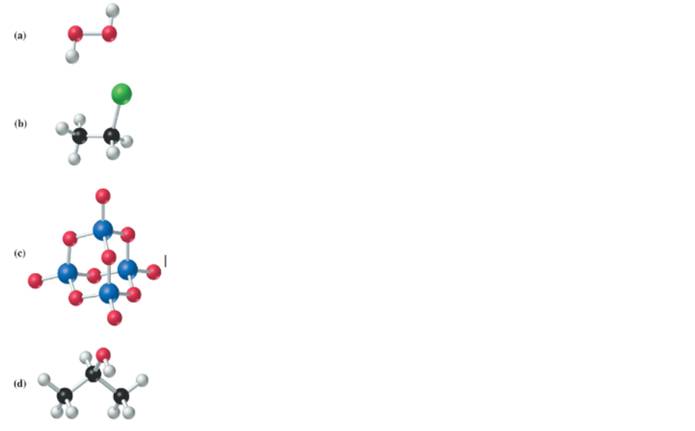
(a)
Interpretation:
The molecular formula for the given ball and stick model of the molecule needs to be determined.
Concept introduction:
The ball and stick model of a chemical substance is defined as a molecular model that displays the 3-D position of the atoms and bonds present between them. In the ball and stick model, the atoms are represented as balls or sphere where different atoms are shown by different colors and bonds are represented by sticks drawn between the atoms. The double and triple bonds are shown by drawing 2 and 3 sticks (curved rod) respectively between the atoms.
Answer to Problem 1E
Explanation of Solution
The given structure is as follows:
According to the color scheme given in figure 3.3 in the book, oxygen atom is represented by red sphere and hydrogen atom by white sphere. Thus, there are 2 oxygen atoms bonded together with single bond and 1 hydrogen atom each.
The molecular formula of the molecule will be
(b)
Interpretation:
The molecular formula for the given ball and stick model of the molecule needs to be determined.
Concept introduction:
The ball and stick model of a chemical substance is defined as a molecular model that displays the 3-D position of the atoms and bonds present between them. In the ball and stick model, the atoms are represented as balls or sphere where different atoms are shown by different colors and bonds are represented by sticks drawn between the atoms. The double and triple bonds are shown by drawing 2 and 3 sticks (curved rod) respectively between the atoms.
Answer to Problem 1E
Explanation of Solution
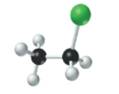
The given structure is as follows:
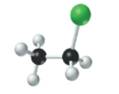
According to the color scheme given in figure 3.3 in the book, carbon atom is represented by black sphere, hydrogen atom by white sphere and chlorine atom by dark green sphere. Thus, there are 2 carbon atoms bonded together. One carbon atom is bonded with 3 hydrogen atoms and other with 2 hydrogen atoms and 1 chlorine atom.
The molecular formula of the molecule will be
(c)
Interpretation:
The molecular formula for the given ball and stick model of the molecule needs to be determined.
Concept introduction:
The ball and stick model of a chemical substance is defined as a molecular model that displays the 3-D position of the atoms and bonds present between them. In the ball and stick model, the atoms are represented as balls or sphere where different atoms are shown by different colors and bonds are represented by sticks drawn between the atoms. The double and triple bonds are shown by drawing 2 and 3 sticks (curved rod) respectively between the atoms.
Answer to Problem 1E
Explanation of Solution
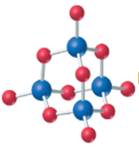
The given structure is as follows:
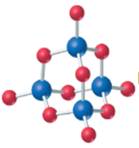
According to the color scheme given in figure 3.3 in the book, phosphorus atom is represented by dark blue sphere and oxygen atom with red sphere. Thus, there are 4 phosphorus atoms and 10 oxygen atoms. The molecular formula of the molecule will be
(d)
Interpretation:
The molecular formula for the given ball and stick model of the molecule needs to be determined.
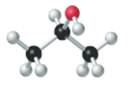
Concept introduction:
The ball and stick model of a chemical substance is defined as a molecular model that displays the 3-D position of the atoms and bonds present between them. In the ball and stick model, the atoms are represented as balls or sphere where different atoms are shown by different colors and bonds are represented by sticks drawn between the atoms. The double and triple bonds are shown by drawing 2 and 3 sticks (curved rod) respectively between the atoms.
Answer to Problem 1E
Explanation of Solution
The given structure is as follows:
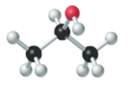
According to the color scheme given in figure 3.3 in the book, carbon atom is represented by black sphere, hydrogen atom by white sphere and oxygen atom by red sphere. Thus, there are 3 carbon atoms. The 2 carbon atoms are attached with 3 hydrogen atoms each and 1 carbon atom is attached with 2 hydrogen atoms and 1oxygen atom. The molecular formula of the molecule will be
(e)
Interpretation:
The molecular formula for the given ball and stick model of the molecule needs to be determined.
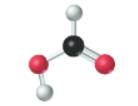 Concept introduction:
Concept introduction:
The ball and stick model of a chemical substance is defined as a molecular model that displays the 3-D position of the atoms and bonds present between them. In the ball and stick model, the atoms are represented as balls or sphere where different atoms are shown by different colors and bonds are represented by sticks drawn between the atoms. The double and triple bonds are shown by drawing 2 and 3 sticks (curved rod) respectively between the atoms.
Answer to Problem 1E
Explanation of Solution
The given molecule is as follows:
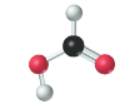
According to the color scheme given in figure 3.3 in the book, carbon atom is represented by black sphere, hydrogen atom by white sphere and oxygen atom by red sphere. The 2 curved rods between 1 carbon and 1 oxygen atom represents the double bond. Thus, there is 1 carbon atom bonded with 1 oxygen atom via double bond, 1 hydrogen atom via single bonds and 1 oxygen atom via single bond which is further attached with another hydrogen atom. The molecular formula of the molecule will be
Want to see more full solutions like this?
Chapter 3 Solutions
EBK GENERAL CHEMISTRY
Additional Science Textbook Solutions
Organic Chemistry
Chemistry & Chemical Reactivity
Living By Chemistry: First Edition Textbook
Organic Chemistry
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry: Structure and Properties
- 2.37 Why are empirical formulas preferred for describing polymer molecules?arrow_forwardQuestion #10-please in alphabetical order (according to formulas, not name)arrow_forwardCyclopropane is an interesting hydrocarbon. Instead of havingthree carbons in a row, the three carbons form a ring, asshown in this perspective drawing (see Figure 2.18 for a priorexample of this kind of drawing): Cyclopropane was at one time used as an anesthetic, but its use was discontinued, in part because it is highly flammable.(a) What is the empirical formula of cyclopropane? Howdoes it differ from that of propane? (b) The three carbon atomsare necessarily in a plane. What do the different wedgesmean? (c) What change would you make to the structureshown to illustrate chlorocyclopropane? Are there isomers ofchlorocyclopropane?arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





