
DRAW IT Ø Label a hydrogen bond and a polar covalent bond in the diagram of live water molecules. Is a hydrogen bond a covalent bond? Explain.
To label: The hydrogen bond and covalent bonds in water molecules.
Introduction:
Water is a polar molecule consisting of an oxygen atom and two hydrogen atoms.
Explanation of Solution
Water (H2O) consists of an oxygen atom and two hydrogen atoms. Hydrogen and oxygen share their valence electrons to form a strong bond that known as a covalent bond (Fig.1). As oxygen is more electronegative than hydrogen, the shared electrons in the H-O bond tend to be pulled towards the oxygen atom. There are two regions of partial negative charge on oxygen and a partial positive charge on each hydrogen atom. Thus, the covalent bond between H-O in the water molecule is a polar bond.
Water is a polar molecule due to the difference in electronegativity between O and H atoms. The partial negatively charged oxygen atom of a water molecule is attracted to the partial positively charged hydrogen atom of an adjacent water molecule (Fig.1). This forms the hydrogen bond among different water molecules.
Pictorial representation:
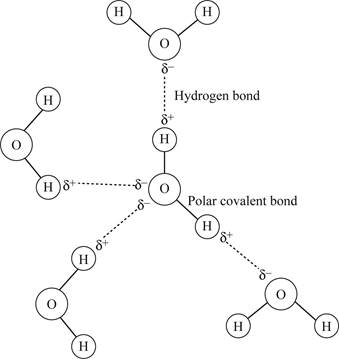
Fig. 1 Interactions among water molecules
To explain: The hydrogen bonds are not covalent bonds.
Introduction: Covalent bonds are formed by the sharing of valence electrons between two atoms. Hydrogen bonds are formed due to the attraction between two adjacent atoms due to partial charges.
Explanation of Solution
Covalent bonds are strong bonds, formed by the sharing of electrons between atoms. Hydrogen bonds do not involve the sharing of valence electrons. They are formed on the basis of attraction due to partial charges on neighboring atoms, and are hence very weak bonds. Therefore, a hydrogen bond is not a covalent bond.
Want to see more full solutions like this?
Chapter 3 Solutions
BIOL 308: BIOL-MSTBIO NEW DSGN ACCESS
Additional Science Textbook Solutions
Human Biology: Concepts and Current Issues
Essentials of Genetics (9th Edition) - Standalone book
Genetics: Analysis and Principles
Biological Science (6th Edition)
Fundamentals of Anatomy & Physiology Plus Mastering A&P with eText - Access Card Package (10th Edition) (New A&P Titles by Ric Martini and Judi Nath)
- Match the terms with their most suitable description. ____ hydrophilic a. protons > electrons ____ atomic number b. number of protons in ____ hydrogen bonds nucleus ____ positive charge c. polar; dissolves easily in ____ negative charge water ____ temperature d. collectively strong ____ pH e. protons < electrons ____ covalent bond f. measure of molecular ____ hydrophobic motion g. water-dreading h. electron sharing i. reflects H+ concentrationarrow_forwardThe partial negative charge at one end of a water molecule is attracted to the partial positive charge of another water molecule. What is this attraction called? (a) a hydrogen bond (b) a van der Waals interaction (c) an ionic bond (d) a covalent bond (e) a hydrophilic bondarrow_forwardExplain in detail why water molecules are polar. Your explanation can be as long as you wish. Your answer must be in your own words. Your answer must include the following terms: Polar, polar covalent bonds, electronegativity, oxygen, hydrogen, partial positive, partial negartive, electrons.arrow_forward
- One of the most important properties of water is that it can form a _______ with other water molecules. This means that water molecules are able to “stick together” to a certain extent. The term for this property is cohesion. A. Ionic bond B. Covalent Bond C. Hydrogen Bondarrow_forwardWhy is it unlikely that two neighboring water moleculeswould be arranged like this?H HH HO Oarrow_forwardDraw a water molecule and a methanol molecule interacting with each other. Label: one polar covalent bond, one nonpolar covalent bond, and one hydrogen bond. Indicate all atoms which have a partial positive or negative charge with the appropriate symbol.arrow_forward
- Which of the following is not a property of water? a. It has a low boiling point compared with other molecules. b. It has a high heat of vaporization. c. Its molecules resist separation, a property called cohesion. d. It has the property of adhesion, the ability to stick to chargedand polar groups in molecules. e. It can form hydrogen bonds to molecules below but not aboveits surface.arrow_forwardFigure 2.12 A pH scale. Here, red dots signify hydrogen ions (H+) and blue dots signify hydroxyl ions (OH). Also shown are the approximate pH values for some common solutions. This pH scale ranges from 0 (most acidic) to 14 (most basic). A change of one unit on the scale corresponds to a tenfold change in the amount of H+ ions. Photos, JupiterImages Corporation. Figure It Out: What is the approximate pH of cola?arrow_forwardWatch this video (http://openstaxcollege.org/l/disaccharide) to observe the formation of a disaccharide. What happens when water encounters a glycosidic bond?arrow_forward
- Atoms share electrons unequally in a(n) ________ bond. a. ionic b. hydrogen c. polar covalent d. nonpolar covalentarrow_forwardAtoms share electrons unequally in an _______ bond. a. ionic c. polar covalent b. hydrogen d. nonpolar covalentarrow_forwardThe water lattice: a. is formed from hydrophobic bonds. b. causes ice to be denser than water. c. causes water to have a relatively low specific heat. d. excludes nonpolar substances. e. is held together by hydrogen bonds that are permanent; that is, they never break and reform.arrow_forward

 Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College





