
Concept explainers
3-38 Complete the chart by writing formulas for the compounds formed:
| Br- | MnO4- | O2- | NO3- | SO42- | PO43- | OH- | |
| Li+ | |||||||
| Ca2+ | |||||||
| Co3+ | |||||||
| K+ | |||||||
| Cu2+ |
Interpretation:
Complete the chart by writing formulas.
Concept Introduction:
The compounds which contain ionic bond, which is a type of bond which formed between positive metal ion and negative, non-metal ions.
Answer to Problem 3.38P
The complete chart is as follows:
Explanation of Solution
Polyatomic ion is a group of covalently bonded nonmetal atoms which has an overall electrical charge.
In general, there are two types of ions: positive metal ions and negative non −metal ions.
When a neutral element gain electrons, the negative charge increases on it and it convert into negative ion. This negative ion is always greater than its parent atom.
Similarly, when a neutral element loss electrons, the negative charge decreases on it and it convert into positive ion. This positive ion is always smaller than its parent atom.
The general rules for writing formula of binary compound are as follows:
- To write the formula of positive ion first.
- The write the formula of negative ions after positive ion.
- Using criss-cross method, the charges of both the ions are interchanged.
- The formula shows the lowest whole-number ratio of both the ions.
For example: the symbols of the magnesium and chloride ions are as follows:
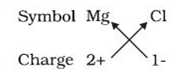
The formula of the ionic compound of these ions is as follows:
The complete chart is as follows:
The chart was completed using the criss-cross method, in which the respective ions are written with their formulas and charge and are then cross multiplied.
Want to see more full solutions like this?
Chapter 3 Solutions
Bundle: Introduction To General, Organic And Biochemistry, 11th + Owlv2, 1 Term (6 Months) Printed Access Card
- 3-56 How many covalent bonds are normally formed by each element? (a)N ( b)F (c)C (d)Br (e)Oarrow_forward3-32 Judging from their relative positions in the Periodic Table, which element in each pair has the larger electronegativity? (a)F or Cl (b) O or S (c) C or N (d) C or Farrow_forward3-70 Draw a Lewis structure of a covalent compound in which oxygen has: (a) Two single bonds and two unshared pairs of electrons (b) One double bond and two unshared pairs of electronsarrow_forward
- 3-31 Why does electronegativity generally increase going from left to right across a row of the Periodic Table?arrow_forward3-18 How many electrons must each atom gain or lose to acquire an electron configuration identical to the noble gas nearest to it in atomic number? (a) Li (b) Cl (c) P (d) Al (e) Sr (f) S (g) Si (h) Oarrow_forward3-41 Describe the structure of sodium chloride in the solid state.arrow_forward
- 3-67 Why does nitrogen have three bonds and one unshared pair of electrons in covalent compounds?arrow_forward3-79 Answer true or false. (a) The letters VSEPR stand for valence-shell electron-pair repulsion. (b) In predicting bond angles about a central atom in a covalent molecule, the VSEPR model considers only shared electron pairs (electron pairs involved in forming covalent bonds). (c) The VSEPR model treats the two electron pairs of a double bond as one region of electron density and the three electron pairs of a triple bond as one region of electron density. (d) In carbon dioxide, OCO, carbon is surrounded by four pairs of electrons and the VSEPR model predicts 109.5° for the OCO bond angle. (e) For a central atom surrounded by three regions of electron density, the VSEPR model predicts bond angles of 120°. (f) The geometry about a carbon atom surrounded by three regions of electron density is described as trigonal planar. (g) For a central atom surrounded by four regions of electron density, the VSEPR model predicts bond angles of 360°/4 = 90°. (h) For the ammonia molecule, NH3, the VSEPR model predicts HNH bond angles of 109.5°. (i) For the ammonium ion, NH4, the VSEPR model predicts HNH bond angles of 109.5°. (j) The VSEPR model applies equally well to covalent compounds of carbon, nitrogen, and oxygen. (k) In water, HOH, the oxygen atom forms covalent bonds to two other atoms, and therefore, the VSEPR model predicts an HOH bond angle of 180°. (l) If you fail to consider unshared pairs of valence electrons when you use the VSEPR model, you will arrive at an incorrect prediction. (m) Given the assumptions of the VSEPR model, the only bond angles it predicts for compounds of carbon, nitrogen, and oxygen are 109.5°, 120°, and 180°.arrow_forward3-46 Which formulas are not correct? For each that is not correct, write the correct formula. (a) Calcium oxide; CaO2 (b) Lithium oxide; LiO (c) Sodium hydrogen phosphate; NaHPO4 (d) Ammonium nitrate; NH4NO3arrow_forward
- 3-87 Consider the molecule boron trffluoride, BF3. (a) Write a Lewis structure for BF3. (b) Predict the FBF bond angles using the VSEPR model. (c) Does BF3 have polar bonds? Is it a polar molecule?arrow_forward2-97 Explain why the Ca3+ ion is not found in chemical compounds.arrow_forward3-68 Draw a Lewis structure of a covalent compound in which nitrogen has: (a) Three single bonds and one unshared pair of electrons (b) One single bond, one double bond, and one unshared pair of electrons (c) One triple bond and one unshared pair of electronsarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
