
Interpretation:
The planar density and packing fraction for the planes (100), (110), and (111) in BCC lithium needs to be calculated and the plane which is close packed needs to be identified.
Concept introduction:
Lithium has a body-centered cubic structure with a lattice parameter of 3.5089
The atomic radius can be calculated as follows:
Here,
Planar density is the ratio of the area of the plane to a number of atoms in a plane.
Answer to Problem 3.77P
Explanation of Solution
For plane (100):
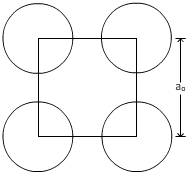
From figure,
For plane (110):
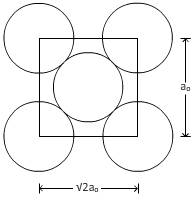
From figure,
For plane(111):
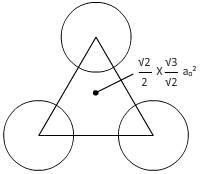
From figure,
Higher the packing fraction, more dense it will be.
From the above calculation, packing fraction of all the planes are:
Hence, plane (110) is highly denser among all the planes because it has a higher value of packing fraction.
Want to see more full solutions like this?
Chapter 3 Solutions
Essentials Of Materials Science And Engineering, Si Edition
 MATLAB: An Introduction with ApplicationsEngineeringISBN:9781119256830Author:Amos GilatPublisher:John Wiley & Sons Inc
MATLAB: An Introduction with ApplicationsEngineeringISBN:9781119256830Author:Amos GilatPublisher:John Wiley & Sons Inc Essentials Of Materials Science And EngineeringEngineeringISBN:9781337385497Author:WRIGHT, Wendelin J.Publisher:Cengage,
Essentials Of Materials Science And EngineeringEngineeringISBN:9781337385497Author:WRIGHT, Wendelin J.Publisher:Cengage, Industrial Motor ControlEngineeringISBN:9781133691808Author:Stephen HermanPublisher:Cengage Learning
Industrial Motor ControlEngineeringISBN:9781133691808Author:Stephen HermanPublisher:Cengage Learning Basics Of Engineering EconomyEngineeringISBN:9780073376356Author:Leland Blank, Anthony TarquinPublisher:MCGRAW-HILL HIGHER EDUCATION
Basics Of Engineering EconomyEngineeringISBN:9780073376356Author:Leland Blank, Anthony TarquinPublisher:MCGRAW-HILL HIGHER EDUCATION Structural Steel Design (6th Edition)EngineeringISBN:9780134589657Author:Jack C. McCormac, Stephen F. CsernakPublisher:PEARSON
Structural Steel Design (6th Edition)EngineeringISBN:9780134589657Author:Jack C. McCormac, Stephen F. CsernakPublisher:PEARSON Fundamentals of Materials Science and Engineering...EngineeringISBN:9781119175483Author:William D. Callister Jr., David G. RethwischPublisher:WILEY
Fundamentals of Materials Science and Engineering...EngineeringISBN:9781119175483Author:William D. Callister Jr., David G. RethwischPublisher:WILEY





