
ORGANIC CHEMISTRY LL W/SSM+CONNECT+KIT
5th Edition
ISBN: 9781259971396
Author: SMITH
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 30, Problem 30.40P
Cationic
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
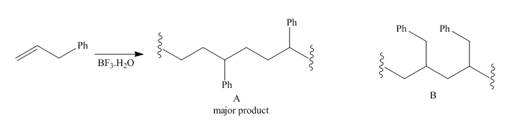
Cationic polymerization of 3-phenylpropene (CH2=CHCH2Ph) affords Aas the major product rather than B. Draw a stepwise mechanism toaccount for this observation.
Styrene derivatives such as A can be polymerized by way of cationic rather than radical intermediates. Cationic polymerization is an example of electrophilic addition to an alkene involving carbocations.
a.) Draw a short segment of the polymer formed by the polymerization ofA.b.) Why does A react faster than styrene (C6H5CH=CH2) in a cationicpolymerization?
Methyl alpha cyanoacrylate (Super Glue) is easily polymerized, even by weak bases. Draw a mechanism for its base-catalyzed polymerization, and explain why this polymerization goes so quickly and easily.
Chapter 30 Solutions
ORGANIC CHEMISTRY LL W/SSM+CONNECT+KIT
Ch. 30 - Prob. 30.1PCh. 30 - Prob. 30.2PCh. 30 - Prob. 30.3PCh. 30 - Draw the mechanism for the radical polymerization...Ch. 30 - Prob. 30.5PCh. 30 - Prob. 30.6PCh. 30 - Prob. 30.7PCh. 30 - Prob. 30.8PCh. 30 - Prob. 30.9PCh. 30 - Prob. 30.10P
Ch. 30 - Prob. 30.11PCh. 30 - Problem 30.12
What polymer is formed by anionic...Ch. 30 - Prob. 30.13PCh. 30 - Prob. 30.14PCh. 30 - Problem 30.15
What polyamide is formed from each...Ch. 30 - Prob. 30.16PCh. 30 - Prob. 30.17PCh. 30 - Prob. 30.18PCh. 30 - Prob. 30.19PCh. 30 - Prob. 30.20PCh. 30 - Prob. 30.21PCh. 30 - Prob. 30.22PCh. 30 - Prob. 30.23PCh. 30 - Prob. 30.24PCh. 30 - Prob. 30.25PCh. 30 - 30.26 Draw the structure of the polymer formed by...Ch. 30 - Prob. 30.27PCh. 30 - Prob. 30.28PCh. 30 - Prob. 30.29PCh. 30 - 30.30 Draw each polymer in Problem 30.29 using the...Ch. 30 - Prob. 30.31PCh. 30 - Prob. 30.32PCh. 30 - Prob. 30.33PCh. 30 - Prob. 30.34PCh. 30 - Prob. 30.35PCh. 30 - Prob. 30.36PCh. 30 - Prob. 30.37PCh. 30 - Prob. 30.38PCh. 30 - 30.39 Draw a stepwise mechanism for the...Ch. 30 - 30.40 Cationic polymerization of 3-phenylpropene ...Ch. 30 - Prob. 30.41PCh. 30 - Prob. 30.42PCh. 30 - 30.43 Although styrene undergoes both cationic and...Ch. 30 - 30.44 Rank the following compounds in order of...Ch. 30 - Prob. 30.45PCh. 30 - Prob. 30.46PCh. 30 - 30.47 Draw a stepwise mechanism for the following...Ch. 30 - 30.48 Draw a stepwise mechanism for the reaction...Ch. 30 - 30.49 Draw the products of each reaction.
a. e....Ch. 30 - Prob. 30.50PCh. 30 - Prob. 30.51PCh. 30 - 30.52 (a) Explain why poly (vinyl alcohol) cannot...Ch. 30 - 30.53 Devise a synthesis of terephthalic acid and...Ch. 30 - Prob. 30.54PCh. 30 - Prob. 30.55PCh. 30 - 30.56 Compound A is a novel poly (ester amide)...Ch. 30 - 30.57 Researchers at Rutgers University have...Ch. 30 - 30.58 Melmac, a thermosetting polymer formed from...Ch. 30 - 30.59 Although chain branching in radical...Ch. 30 - Prob. 30.60P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the starting structure that would lead to this polymer under radical condarrow_forwardIn the presence of H3O+, 2-methylpropene oxide undergoes chain-growth polymerization such that nucleophilic attack occurs at the more substituted end of the epoxide. Draw a stepwise mechanism for this process, and explain this regioselectivity.arrow_forwardThe polymer shown below is synthesized by hydroxide ion-promoted hydrolysis of a copolymer of para-nitrophenyl methacrylate and acrylate. a. Propose a mechanism for the formation of the copolymer. b. Explain why hydrolysis of the copolymer to form the polymer occurs much more rapidly than hydrolysis of para-nitrophenyl acetate.arrow_forward
- Draw a stepwise mechanism for the conversion of acrylonitrile (CH2 = CHC ≡ N) to polyacrylonitrile, –[CH2CHC ≡ N]n–, using butyllithium (BuLi) as the initiator and CO2 as the electrophile to terminate the chain.arrow_forwardA3 Anionic polymerization of styrene is generally known to produce polymers with a narrow molecular weight distribution. Explain the reason for this.arrow_forward(a) Explain why poly(vinyl alcohol) cannot be prepared by the radical polymerization of vinyl alcohol (CH2=CHOH). (b) Devise a stepwise synthesis of poly(vinyl alcohol) from vinyl acetate (CH2=CHOCOCH3). (c) How can poly(vinyl alcohol) be converted to poly(vinyl butyral), a polymer used in windshield safety glass?arrow_forward
- Draw a structural formula of the polymer resulting from base-catalyzed polymerization of each compound. Would you expect the polymers to be optically active? (S)-(+)-lactide is the dilactone formed from two molecules of (S)-(+)-lactic acid.arrow_forwardChain branching occurs in cationic polymerization much as it does in free-radical polymerization. Propose a mechanism to show how branching occurs in the cationic polymerization of styrene. Suggest why isobutylene might be a better monomer for cationic polymerization than styrene.arrow_forwardPoly(vinyl alcohol) is a polymer used to make fibers and adhesives. It is synthesized by hydrolysis or alcoholysis of the polymer obtained from polymerization of vinyl acetate as shown below. a. Why is poly(vinyl alcohol) not prepared by polymerizing vinyl alcohol? b. Is poly(vinyl acetate) a polyester?arrow_forward
- Although many alkenes are readily polymerized by a free radical mechanism, poly(isobutylene) (CAS# 9003-27-4) is often produced via a cationic route. Offer an explanation for this unique behavior of isobutylene. Explain and support your answer.arrow_forwardList the following group of monomers in order of decreasing ability to undergo cationic polymerization.arrow_forward(a) Hard contact lenses, which first became popular in the 1960s, were made by polymerizing methyl methacrylate [CH2=C(CH3)CO2CH3] to form poly(methyl methacrylate) (PMMA). Draw the structure of PMMA. (b) More-comfortable softer contact lenses introduced in the 1970s were made by polymerizing hydroxyethyl methacrylate [CH2=C(CH3)CO2CH2CH2OH] to form poly(hydroxyethyl methacrylate) (poly-HEMA). Draw the structure of poly-HEMA. Because neither polymer allows oxygen from the air to pass through to the retina, newer contact lenses that are both comfortable and oxygen-permeable have now been developed.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY