
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.4, Problem 3.30P
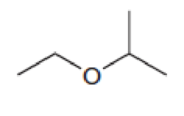
Predict the multiplicity of each signal in the expected proton NMR spectrum of each of the following compounds:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Determine the multiplicity of each signal in the expected 1H and 13C NMR spectrum of the following compound.
Predict the number of signals in a proton-decoupled 13C-NMR spectrum of each compound
Determine the multiplicity and predict the chemical shifts of each signal in the expected 1H NMR spectrum of the following compound.
Chapter 3 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 3.1 - Prob. 3.2PCh. 3.1 - Prob. 3.3PCh. 3.1 - Prob. 3.4PCh. 3.1 - Prob. 3.5PCh. 3.1 - Prob. 3.6PCh. 3.1 - Prob. 3.7PCh. 3.1 - Prob. 3.8PCh. 3.1 - Prob. 3.9PCh. 3.1 - Prob. 3.10PCh. 3.1 - If you look at your answers to the previous...
Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Prob. 3.19PCh. 3.3 - Prob. 3.21PCh. 3.3 - Prob. 3.22PCh. 3.3 - Prob. 3.23PCh. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.9 - Prob. 3.43PCh. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Prob. 3.48PCh. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
A 54 cm2 ice cube at 25.0 °C is added to a thermally insulated container with 400.0 mL H2O(l) at 32.0 °C. What ...
General Chemistry: Principles and Modern Applications (11th Edition)
For each of the following molecules, (1) draw the correct Lewis structure; (2) label each polar covalent bond w...
General, Organic, and Biological Chemistry (3rd Edition)
The balance chemical equation between sodium phosphate and cobalt (III) nitrate to form purple precipitate need...
Chemistry: Matter and Change
Practice Problem 7.10 When cis-1-bromo-4-tert-butylcyclohexane is treated with sodium ethoxide in ethanol, it r...
Organic Chemistry
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How many 1H-NMR signals/peaks would you expect to see for the following molecule?arrow_forwardHow many peaksPredict the number of peaks that you would expect in the proton decoupled 13C spectrum and 1H spectrum of each of the following compoundsarrow_forwardPredict the chemical shift and spiting pattern of each of the signals in the 1H NMR spectrum and sketch the spectrum of compound A above.arrow_forward
- What is the molecular formula structure and its proton environment of these two spectrum?arrow_forwardEstimate the chemical shift expected "iii" in the H-NMR spectrum of the molecule shown below.arrow_forwardPredict the approximate chemical shift of the indicated peaks in the C-NMR spectrum.arrow_forward
- Which compound gives a signal in the 1H-NMR spectrum with a larger chemical shift, furan or cyclopentadiene? Explain.arrow_forwardProvide the chemical shifts, integration (or ratio), and multiplicity for the signals in the 1H NMRspectrum and draw the expected 1H NMR spectrum for each of the following compounds:arrow_forwardV.) A compound with molecular formula C8H10O has the following proton NMR spectrum. Determine the number of protons giving rise to each signal (a, b, c, and d). (SEE PICTURE ATTACHED).arrow_forward
- Label each set of chemically equivalent protons, using a for the set that will be at the lowest frequency in the 1H NMR spectrum, b for the next lowest,and so on. Indicate the multiplicity of each signal.arrow_forwardWhat is the number of signals that will be observed for each of the following molecules in the 1H NMR spectrum?arrow_forward07) How could 1H and 13C NMR distinguish between substances in each of the following pairs?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY