
Concept explainers
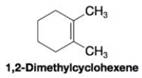
When 1,2-dimethylcyclohexene is allowed to react with hydrogen in the presence of a platinum catalyst, the product of the reaction is a cycloalkane that has a melting point of –50 °C and a boiling point of 130 °C (at 760 torr).
(a) What is the structure of the produce of this reaction?
(b) Consult an appropriate resource (such as the web or a CRC handbook) and tell which stereoisomer it is.
(c) What does this experiment suggest about the mode of addition of hydrogen to the double bond?
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Organic Chemistry (9th Edition)
Chemistry: The Central Science (13th Edition)
Living By Chemistry: First Edition Textbook
Chemistry: The Central Science (14th Edition)
Organic Chemistry As a Second Language: Second Semester Topics
- (b) (1-chloromethyl)cyclopentane, CeH11CI reacts with aqueous sodium hydroxide, NaOH to produce a primary alcohol AA. When CeH11Cl is added with magnesium, Mg in ether, an organometallic compound BB is formed. When compound BB reacts with ethanal, CH3CHO, a secondary alcohol cCis formed. The molecular structure of CeH11Cl is given below. (1-klorometil)siklopentana, CoH1,CI bertindak balas dengan akues natrium hidroksida, NaOH bagi menghasilkan satu alkohol primer AA. Apabila CeH11CI ditambah dengan magnesium, Mg dalam eter, sebatian organologam BB terbentuk. Apabila sebatian BB bertindak balas dengan etanal, CH;CHO, satu alkohol sekunder CC dihasilkan. Struktur molekul bagi CsH11CI diberikan di bawah. .CI (1-chloromethyl)cyclopentane (i) Draw the structural formula for compound AA through Cc. Lukiskan formula struktur bagi sebatian AA sehingga C. (ii) What is the type of reaction to produce compound AA? Apakah jenis tindak balas untuk menghasilkan sebatian AA?arrow_forwardThe melting points and boiling points of two isomeric alkanes are as follows: CH3(CH2)6CH3, mp = -57 °C and bp = 126 °C; (CH3)3CC(CH3)3, mp = 102 °C and bp = 106 °C. (a) Explain why one isomer has a lower melting point but higher boiling point. (b) Explain why there is a small difference in the boiling points of the two compounds, but a huge difference in their melting points.arrow_forward(a) What structural feature is associated with each type of hy-drocarbon: an alkane; a cycloalkane; an alkene; an alkyne? (b) Give the general formula for each type .(c) Which hydrocarbons are considered saturated?arrow_forward
- Which of the following alkenes can exist as cis-trans isomers? Write their structures. (a) CH2=CHCH2CH3 (c) CH2=C(CH3)2 (b) CH3CH=CHCH3 (d) CH3CH2CH=CHCIarrow_forward(d) This structure is that of an alcohol with the hydroxyl group on the third C atom of a five-carbon chain.The compound is called pentan-3-olarrow_forwardWrite the reagent or draw structures of the starting material or organic product(s) in the following reactions. If more than one product is formed, identify the major product where possible. (a) (b) HO OH OH H2SO4 ? Cl₂ ? FeCl3arrow_forward
- The skeletal line formula for a branched alkene is shown below. (i) What is the molecular formula of this compound? (ii) How many carbon atoms are in the longest chain, ignoring the double bond? (iii) What is the longest chain incorporating both carbons of the double bond? (iv) How many substituents are on this chain? (v) Give the IUPAC name for this compound. [6]arrow_forwardWrite the bond line formula of the following compounds: (a) 4-methyl-2-hexene, two geometrical (stereoisomers) isomers (b) 3-fluoro-2-methylheptanol (3-fluoro-2-methylheptan-1-ol) (c) 4-methyl-hex-1-yn-3-olarrow_forwardAll about Alkene, Alkyne and Alkyl halides(1) Write a complete chemical equation showing reactants, products, and catalysts needed (if any) for the following reaction and (2) Draw and name the organic compound found in every reaction. (A) Reaction of cis-3,3-Dimethyl-4-propylocta-1,5-diene with two mole of HBr(B) Reaction of trans-1-Bromo-3-chlorocyclopentane with potassium hydroxide(C) Formation of Gilman reagent using isopropyl bromidearrow_forward
- Which of the following cycloalkanes are capable of geometric (cis-trans) isomerism?Draw the cis and trans isomers.(a) 3-ethyl-1,1-dimethylcyclohexane (b) 1-ethyl-3-methylcycloheptane(c) 1-ethyl-3-methylcyclopentane (d) 1-cyclopropyl-2-methylcyclohexanearrow_forwarda) Draw structures for the following compounds. (i) (ii) (iii) 3-ethyl-4-methylhexane 4-tert-butyl-2-methylheptane 3-methylpent-1-ene (b) Provide the IUPAC names for the following (i) (ii) (CH3)3C(CH2)3CH(CH₂CH3)CH₂CH₂CH3arrow_forwardPropose a structural formula for the product(s) when each of the following alkenes is treated with H2O/H,SO4. Why are two products formed in part (b), but only one in parts (a) and (c)? (a) 1-Hexene gives one alcohol with a molecular for- mula of C,H140. (b) 2-Hexene gives two alcohols, each with a molecu- lar formula of C,H140. (c) 3-Hexene gives one alcohol with a molecular for- mula of C6H140.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
