
Fuel oils contain primarily organic compounds and sulfur. The molar composition of the organic fraction of a fuel oil may be represented by the formula
the mass fraction of sulfur in the fuel is
As (kg S/kg fuel); and the percentage excess air, Pn, is defined in terms of the theoretical air required to burn only the carbon and hydrogen in the fuel.
- For a certain high-sulfur No. 6 fuel oil, p = 0.71, 2, SO2, and H2O and expressing the SO2 fraction as ppm (mol SO2/I()6mol dry gas).
- Create a spreadsheet to calculate the mole fractions of the stack gas components on a dry basis for specified values of p, q, r, as, and PXi. The output should appear as follows:
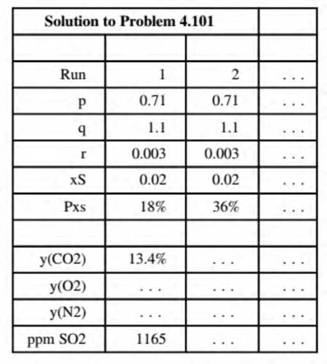
(Rows below the last one shown can be used to calculate intermediate quantities.) Execute enough runs (including the two show n above) to determine the effect on the stack gas composition of each of the five input parameters. Then for the values of p, q, r. and As given in Part (a), find the minimum percentage excess air needed to keep the dry-basis SO2 composition below 700 ppm. (Make this the last run in the output table.)
You should find that for a given fuel oil composition, increasing the percentage excess air decreases the SO2 concentration in the stack gas. Explain w hy this should be the case.
- Someone has proposed using the relationship betw een PX5 and ppm SO2 as the basis of a pollution control strategy. The idea is to determine the minimum acceptable concentration of SO2 in the stack gas, then run with the percentage excess air high enough to achieve this value. Give several reasons w hy this is a poor idea.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Elementary Principles Of Chemical Processes
Additional Engineering Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Elementary Surveying: An Introduction To Geomatics (15th Edition)
Engineering Mechanics: Statics
Mechanics of Materials
Applied Statics and Strength of Materials (6th Edition)
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





