
(Sample Problems 4.1 and 4.2)
4.1 What two factors cause water to be polar?
Interpretation: Two factors that cause polarity in
Concept introduction: Solution is made by two portions, solute, and solvent. Solute represents substance present in small quantity while solvent represents substance that dissolves solute.
Polarity in molecule is generated if it has unequal distributed electron density between bonds. Polar molecule has partial charges on atoms of the molecule. The general representation of polar molecule is as follows:
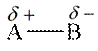
Here, B has high electronegative value than A.
Explanation of Solution
Water molecule has bend shape. In this shape, oxygen present as central atom and two hydrogen atoms are present as side atom. Structure of

Bond
Want to see more full solutions like this?
Chapter 4 Solutions
Principles of General Chemistry
- What volume of 0.687 M perchloric acid solution (in mL) is needed to neutralize 50.00 mL of 0.0347 M sodium hydroxide? (Solution stoichiometry)arrow_forwardWhat volume of 0.547 M sulfuric acid solution (in mL) is needed to neutralize 50.00 mL of 0.0812 M sodium hydroxide? (Using solution stoichiometry)arrow_forwardare all acids electrolytes in water? true or falsearrow_forward
- What dissolved species are present in a solution of KCN?arrow_forwarda. What products will be produced by the acid/base neutralization reaction between sulfuric acid (H2SO4) and potassium hydroxide (KOH)? assume that H2SO4 is a strong diprotic acid, it donates two H+ ions that react with two moles of base. b. Write balanced molecular, total ionic, and net ionic equations for the acid/base neutralization reaction between sulfuric acid (H2SO4) and potassium hydroxide (KOH).arrow_forwardWhy is it a misconception to classify the addition of acids and bases as a "neutralization" reaction?arrow_forward
- Complete the following instructions for the problems below I. Translate the following reactants from chemical names into chemical formulas.II. Predict the products for the reaction (these will be exchange reactions!).III. Balance the reaction.IV. If the reaction is a Precipitation Reaction, write out the complete and net ionicequations for the reaction.V. If the reaction is an Acid-Base Reaction, label the acid, base, the appropriate products,and the net ionic equation.a. Ammonium iodide and silver nitrateb. Potassium carbonate and aluminum sulfatec. Chromic acid and lithium hydroxided. Hydrobromic acid and sodium hydroxidearrow_forwardWhat happens to sodium carbonate when it dissolves in water?arrow_forwardIn a solution, what are electrolytes and non-electrolytes?arrow_forward
- In an accident, a solution containing 745 grams of nitric acid was spilled. To clean it up 900 grams of Sodium Carbonate was quickly spread over the area and carbon dioxide was released by the reaction. Was sufficient Sodium Carbonate used to neutralize all the acid?.arrow_forwardHow many milliliters of 0.837 M H2SO 4 would be required to exactly neutralize 699 mL of 0.532 M NaOH?arrow_forwardHow to write ionic and net ionic equation step by step?in details?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





