
4-44 Answer true or false.
(a) The mole is a counting unit, just as a dozen is acounting unit.
(b)
(c) Avogadro’s number, to three significant figures, is 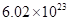 formula units per mole.
formula units per mole.
(d) 1 mol of H2O contains 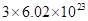 formula units.
formula units.
(e) 1 mol of H2O has the same number of molecules as 1 mol of H2O2.
(f) The molar mass of a compound is its formula weight expressed in amu.
(g) The molar mass of H2O is 18 g/mol.
(h) 1 mol of H2O has the same molar mass as 1 mol of H2O2.
(i) 1 mol of ibuprofen, C13H18O2, contains 33 mol of atoms.
(j) To convert moles to grams, multiply by Avogadro’s number.
(k) To convert grams to moles, divide by molar mass.
(l) 1 mol of H2O contains 1 mol of hydrogen atoms and one mol of oxygen atoms.
(m) 1 mol of H2O contains 2 g of hydrogen atoms and 1 g of oxygen atoms.
(n) 1 mole of H2O contains 18.06 X 10 atoms.
Trending nowThis is a popular solution!

Chapter 4 Solutions
Bundle: Introduction to General, Organic and Biochemistry, 11th + OWLv2, 4 terms (24 months) Printed Access Card
- 4-89 If 7.0 kg of is added to 11.0 kg of to form which reactant is in excess?arrow_forward4-91 Assume that the average red blood cell has a mass of 2 × 10-8 g and that 20% of its mass is hemoglobin (a protein whose molar mass is 68,000). How many molecules of hemoglobin are present in one red blood cell?arrow_forward4-21 The brilliant white light in some firework displays is produced by burning magnesium in air. The magne sium reacts with oxygen in the air to form magnesium oxide. Write a balanced equation for this reaction.arrow_forward
- 4-20 Calcium oxide is prepared by heating limestone (calcium carbonate, CaCO3) to a high temperature, at which point it decomposes to calcium oxide and carbon dioxide. Write a balanced equation for this preparation of calcium oxide.arrow_forward4-52 The molecular weight of hemoglobin is about 68,000 amu. What is the mass in grams of a single molecule of hemoglobin?arrow_forward4-25 In the chemical test for arsenic, the gas arsme, AsH3, is prepared. When arsine is decomposed by heating, arsenic metal deposits as a mirror-like coating on the surface of a glass container and hydrogen gas, H2, is given off. Write a balanced equation for the decomposition of arsine.arrow_forward
- 4-56 Magnesium reacts with sulfuric acid according to the following equation. How many moles of lI2 are produced by the complete reaction of 230. mg of Mg with sulfuric acid?arrow_forward4-57 Chloroform, CHCl3, is prepared industrially by the reaction of methane with chlorine. How many grams of Cl2 are needed to produce 1.50 moles of chloroform?arrow_forward4-54 Answer true or false. (a) Stoichiometry is the study of mass relationships in chemical reactions. (b) To determine mass relationships in a chemical reaction, you first need to know the balanced chemical equation for the reaction. (c) To convert from grams to moles and vice versa, use Avogadro’s number as a conversion factor. (d) To convert from grams to moles and vice versa, use molar mass as a conversion factor. (e) A limiting reagent is the reagent that is used up first. (f) Suppose a chemical reaction between A and B requires 1 mol of A and 2 mol of B. If 1 mol of each is present, then B is the limiting reagent. (g) Theoretical yield is the yield of product that should be obtained according to the balanced chemical equation. (h) Theoretical yield is the yield of product that should be obtained if all limiting reagent is con verted to product. (j) Percent yield is the number of grams of product divided by the number of grams of the limiting reagent times 100. (j) To calculate percent yield, divide the mass of product formed by the theoretical yield and multiply by 100.arrow_forward
- Write an equation from the following description: reactants are gaseous NH3 and O2, products are gaseous NO2 and liquid H2O, and the stoichiometric coefficients are 4, 7, 4, and 6, respectively.arrow_forward4-53 A typical deposit of cholesterol, C27H46O, in an artery might have a mass of 3.9 mg. How many mol ecules of cholesterol are in this mass?arrow_forward4-61 In photosynthesis, green plants convert CO2 and H2O to glucose, C6H12O6. How many grams of CO2are required to produce 5.1 g of glucose?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





