
Organic Chemistry&mod Mstg Etx Vp Ac Pkg
1st Edition
ISBN: 9780134466729
Author: Bruice
Publisher: Pearson Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 72P
Assign relative priorities to each set of substituents:
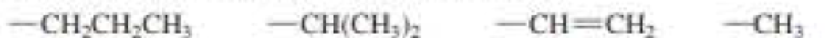

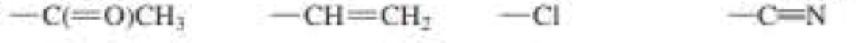
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
a.) Draw B as a hexagon with wedges and dashed wedges to show the stereochemistry of substituents.b.) Draw a stereoisomer of A as a hexagon using wedges and dashed wedges to show the orientation of substituents.
Assign relative priorities to each set of substituents:
a. -CH2CH2CH3 -CH(CH3)2 -CH = CH2 -CH3
b. - CH2NH2 -NH2 - OH -CH2OH
c. -C(=O)CH3 - CH =CH2 -Cl -C=N
Rank the following groups in order of decreasing priority.
−CH=CH2, −CH3, −C≡CH, −H
Chapter 4 Solutions
Organic Chemistry&mod Mstg Etx Vp Ac Pkg
Ch. 4.1 - Prob. 2PCh. 4.1 - Prob. 3PCh. 4.1 - Prob. 4PCh. 4.1 - Prob. 5PCh. 4.1 - Which of the roll owing compounds have a dipole...Ch. 4.2 - Draw and label the E and Z isomers for each of the...Ch. 4.2 - Assign relative priorities to each set of...Ch. 4.2 - Tamoxifen slows the growth of some breast tumors...Ch. 4.2 - Prob. 10PCh. 4.2 - Name each of the following:
Ch. 4.2 - Draw the Z isomer of an alkene that has a CH3 and...Ch. 4.3 - Prob. 13PCh. 4.4 - Prob. 14PCh. 4.5 - Prob. 16PCh. 4.6 - Prob. 17PCh. 4.7 - Prob. 18PCh. 4.8 - Prob. 20PCh. 4.8 - Prob. 22PCh. 4.8 - Prob. 23PCh. 4.8 - Prob. 24PCh. 4.8 - Draw a perspective formula for each or the...Ch. 4.8 - Prob. 27PCh. 4.9 - Prob. 28PCh. 4.9 - What is the configuration of the following...Ch. 4.9 - Prob. 32PCh. 4.10 - Prob. 33PCh. 4.10 - (S)-(+)-Monosodium glutamate (MSG) is a flavor...Ch. 4.11 - Prob. 35PCh. 4.11 - Prob. 36PCh. 4.12 - Prob. 38PCh. 4.12 - Prob. 39PCh. 4.12 - The stereoisomer of cholesterol found in nature is...Ch. 4.12 - Prob. 41PCh. 4.13 - 1-Bromo-2-methylcyclopentane has four pairs of...Ch. 4.13 - Prob. 43PCh. 4.13 - Draw all possible stereoisomers for each of the...Ch. 4.13 - Prob. 45PCh. 4.13 - Of all the possible cyclooctanes that have one...Ch. 4.13 - Prob. 47PCh. 4.13 - Prob. 48PCh. 4.14 - Which of the following compounds has a...Ch. 4.14 - Draw all the stereoisomers for each of the...Ch. 4.15 - Prob. 52PCh. 4.15 - Name the isomers you drew in Problem 52.Ch. 4.15 - Chloramphenicol is a broad-spectrum antibiotic...Ch. 4.15 - Draw a perspective formula for each of the...Ch. 4.15 - Name the following:Ch. 4.15 - Prob. 57PCh. 4.15 - Prob. 59PCh. 4.15 - Convert the perspective formula to a skeletal...Ch. 4.15 - Prob. 62PCh. 4.16 - Prob. 63PCh. 4.17 - Limonene exists as two different stereoisomers....Ch. 4 - a. Draw three constitutional isomers with...Ch. 4 - Prob. 65PCh. 4 - Prob. 66PCh. 4 - Which of the following has an asymmetric center?...Ch. 4 - Prob. 68PCh. 4 - Prob. 69PCh. 4 - Prob. 70PCh. 4 - Prob. 71PCh. 4 - Assign relative priorities to each set of...Ch. 4 - Prob. 73PCh. 4 - Which of the following are optically active?Ch. 4 - Prob. 75PCh. 4 - Name the following:Ch. 4 - Which of the following has an achiral...Ch. 4 - Using skeletal structures, draw the stereoisomers...Ch. 4 - Prob. 79PCh. 4 - Citrate synthase, one of the enzymes in the series...Ch. 4 - Prob. 81PCh. 4 - Prob. 82PCh. 4 - Prob. 83PCh. 4 - Prob. 84PCh. 4 - Prob. 85PCh. 4 - Prob. 86PCh. 4 - Prob. 87PCh. 4 - Prob. 88PCh. 4 - Prob. 89PCh. 4 - a. Draw all the isomers with molecular formula...Ch. 4 - Prob. 91PCh. 4 - Prob. 92PCh. 4 - Draw structures for the following: a....Ch. 4 - For each of the following structures, draw the...Ch. 4 - Prob. 95PCh. 4 - Prob. 96PCh. 4 - Prob. 97PCh. 4 - a. Using the wedge-and-dash notation, draw the...Ch. 4 - Prob. 99PCh. 4 - Prob. 100PCh. 4 - Prob. 101PCh. 4 - a. Draw the two chair conformers for each of the...Ch. 4 - Prob. 103PCh. 4 - Is the following compound optically active?Ch. 4 - Prob. 105P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Conformer E is a (cis/trans) isomer. A conformational ring flip of conformer E will yield conformer F in which the substituent on carbon 1 will be (axial/equatorial/neither) and the substituent on carbon 3 will be (axial/equatorial/neither). Conformer (E/F) will be more stable.arrow_forwardRank the set of substituents below in order of priority according to the Cahn-Ingold-Prelog sequence rules.1 = highest priority.(a) -ch3 (b) -br (c) -h (d) -iarrow_forwardFor the given ee values, calculate the percentage of each enantiomer present.a. 90% eeb. 99% eec. 60% eearrow_forward
- How do I know if the substituent is an EWG or EDGarrow_forwardDraw a Newman projection looking down the C1-C2 bond of 1-chloro-2-methylcyclohexane in such a way that the two substituents are gauche to each other. Then draw another Newman projection in which the two substituents are anti to each other.arrow_forwardGlucose is a simple sugar with five substituents bonded to a sixmembered ring.a.Using a chair representation, draw the most stable arrangement of these substituents on the six-membered ring. b.Convert this representation to one that uses a hexagon with wedges and dashed wedges. c.Draw a constitutional isomer of glucose. d.Draw a stereoisomer that has an axial OH group on one carbon.arrow_forward
- a) Sighting down the C3-C4 bond, draw the gauche (60 degrees) and anti (180 degrees) Newman projections of 2,4-dimethylhexane. b) Circle the conformation that you drew that is lower energy.arrow_forwardCan you help me draw the enantiomer saw-horse conformation of this Newman projection?arrow_forwardRank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Clarrow_forward
- (a) (1R,2R)-1,2-dibromocyclohexane, draw any enantiomer.arrow_forwardFor rotation about the C3-C4 bond of 2-chlorohexane: a) Draw a Newman projection showing the most stable conformation. b) Draw a Newman projection showing the least stable conformation.arrow_forwardAssign relative priorities to each set of substituents: a. ¬Br ¬I ¬OH ¬CH3b. ¬CH2CH2OH ¬OH ¬CH2Cl ¬CH“CH2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License