
(a)
Interpretation:
The three dimensional representation for
Concept introduction:
Electrons in an atom are present in particular orbitals. Sublevels contain orbitals which possess equivalent energy. Maximum two electrons are present in each orbital. The electrons present in
Answer to Problem 81E
The three dimensional representation for
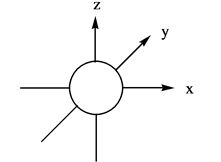
Explanation of Solution
The energy level possesses a sublevel that contains orbital. The sublevels are named as
The three dimensional representation for

Figure 1
The three dimensional representation for
(b)
Interpretation:
The three dimensional representation for
Concept introduction:
Electrons in an atom are present in particular orbitals. Sublevels contain orbitals which possess equivalent energy. Maximum two electrons are present in each orbital. The electrons present in
Answer to Problem 81E
The three dimensional representation for
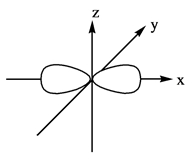
Explanation of Solution
The energy level possesses a sublevel that contains orbital. The sublevels are named as
The three dimensional representation for

Figure 2
The three dimensional representation for
(c)
Interpretation:
The three dimensional representation for
Concept introduction:
Electrons in an atom are present in particular orbitals. Sublevels contain orbitals which possess equivalent energy. Maximum two electrons are present in each orbital. The electrons present in
Answer to Problem 81E
The three dimensional representation for
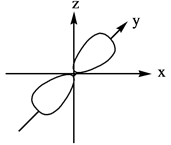
Explanation of Solution
The energy level possesses a sublevel that contains orbital. The sublevels are named as
The three dimensional representation for

Figure 3
The three dimensional representation for
(d)
Interpretation:
The three dimensional representation for
Concept introduction:
Electrons in an atom are present in particular orbitals. Sublevels contain orbitals which possess equivalent energy. Maximum two electrons are present in each orbital. The electrons present in
Answer to Problem 81E
The three dimensional representation for
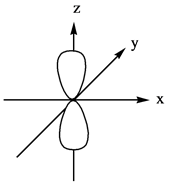
Explanation of Solution
The energy level possesses a sublevel that contains orbital. The sublevels are named as
The three dimensional representation for

Figure 4
The three dimensional representation for
Want to see more full solutions like this?
Chapter 4 Solutions
EP INTRODUCTORY CHEM.-MOD.MASTERINGCHEM
- Give the possible values of a. the principal quantum number, b. the angular momentum quantum number, c. the magnetic quantum number, and d. the spin quantum number.arrow_forwardIn what main group(s) of the periodic table do elements have the following number of half-filled p-orbitals in the outermost principal energy level? (a) 0 (b) 1(c) 2(d) 3arrow_forwardWhat is the physical significance of the value of 2 at a particular point in an atomic orbital?arrow_forward
- For the following pairs of orbitals, indicate which is higher in energy in a many-electron atom. (a) 3s or 2P (b) 4s or 4d (c) 4f or 6s (d) 1s or 2sarrow_forwardHow does the wavelength of a fast-pitched baseball compare to the wavelength of an electron traveling at 110 the speed of light? What is the significance of this comparison? See Example 2-3.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning  Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





