
Concept explainers
(a)
Interpretation:
The following ball- and -stick model of 3-methyl-2-hexenoic acid should be converted into a Lewis structure.
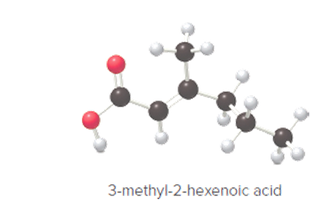
Concept introduction:
- Lewis structures are the diagrams that show the bonding between the atoms of the molecules and existing lone pairs of electrons.
- Bonding electrons are those electrons which are shared between the atoms resulting in the formation of bond.
- Non-bonding electrons are the valence electrons of the atom which are not shared with another atom.
(b)
Interpretation:
The shape around each carbon atom should be determined.
Concept introduction:
The model which is used in chemistry to determine the geometry of individual molecules with the help of the number of electron pairs surrounded by central atoms is known as Valence shell electron pair repulsion (VSEPR) theory.
(c)
Interpretation:
The polar bonds present in 3-methyl-2-hexenoic acid should be labeled.
Concept introduction:
A polar bond is a covalent bond between two atoms where the electrons forming the bond are unequally distributed. This causes the molecule to have a slight electrical dipole moment where one end is slightly positive and the other is slightly negative.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- Write a Lewis structure for each of the following molecules. H2CO (carbon is central) Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardDraw a Lewis structure for the given skeletal molecule. Note: Multiple bonds and lone pairs are not shown in the skeletal molecule. H H H H H N O 0-C N-C-C-ċ-OH C-C нн H-O H How many lone pairs of electrons are found in the Lewis structure of your molecule?arrow_forwardHelparrow_forward
- Predicting whether molecules are polar or onpo Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. For example, if the molecule were HCl and you decided the hydrogen atom was closest to the negative side of the molecule, vou'd enter "H" in the last column of the table. atom closest to polar or nonpolar? molecule or polyatomic ion negative side O polar HCN O nonpolar O polar SIF, nonpolar polar HCL O nonpolar Explanation Check © 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy Accessibility Ov I 11:31 acer ied ned pag din heme page up esc 2$ % & %23 * backspace delete 3 5 7 8 9 tab e y u Potesarrow_forwardComplete the following Lewis structure by adding in missing lone pairs and pi bonds. Assume all atoms are neutral (having no formal charge). Do not add additional atoms to the structure.arrow_forwardDraw a Lewis structure for the compound whose skeletal structure is provided to you below. Don't forget to draw in the H atoms! CH₂ HC CH 11 HC — CH, - Draw the Lewis structure by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forward
- Write a Lewis structure for each of the following molecules. H2O2 Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardPick the approximate bond angle indicated of the molecule with the Lewis structure below. All atoms except H have an octet of electrons. Lone pairs are not indicated. H C-H H-C-0-C Harrow_forwardDraw the Lewis Structure for NH2CH2CO2H. Now answer the following questions based on your Lewis structure: (Enter an integer value only.) # single bonds in the entire molecule # double bonds in the entire molecule # lone pairs in the entire moleculearrow_forward
- complete the following lewis structure (draw preferred) and fill in the table. show formal charges on lewis structure.arrow_forwardDraw a Lewis structure that obeys the octet rule for each of the following ions. Obeying the octet rule may mean that the structure you draw is not the most stable or ideal for the given molecule. Assign formal charges to each atom. ClO3− Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons. Show the formal charges of all nonhydrogen atoms.arrow_forwardDecide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. For example, if the molecule were Ha and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter ":" in the last column of the table. molecule or polyatomic ion polar or nonpolar? atom closest to negative side polar HI O nonpolar O polar H,S O nonpolar O polar CH4 O nonpolararrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

