
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
Which diagram best represents the ions in solution at the equivalence point in the titration of
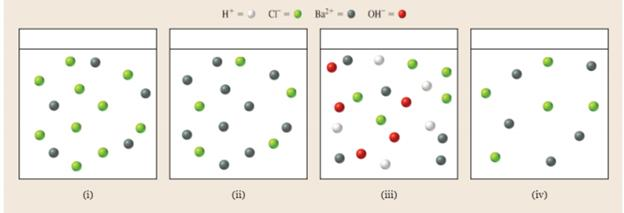
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
CHEMISTRY LOOSELEAF TEXT W/CONNECT >IP
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
General, Organic, & Biological Chemistry
Chemistry: Structure and Properties (2nd Edition)
Principles of General, Organic, Biological Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- Rush. Determine the pH when the following amounts of titrant is added in the following titration: Titration of 75.0 mL of 0.20 M methylamine (CH3NH2, Kb = 4.4 x 10 ^-4) with 0.25 M HCl. a.) What is the pH of the solution after addition of 0.00 mL acid titrant? b.) 10.00 mL titrant? c.) 30.00 mL titrant? d.) 60.00 mL titrant? e.) 61.00 mL titrant?arrow_forwardA buffer is made up of equal volumes (451.8 mL of each) of 0.812 M A H and 0.885 M A minus. 24.51 mL of 0.712 M HCl is added to the buffer. How many moles of A H are present after the addition of HCl? Please use correct significant figures. Answer: (0.384) (mol)arrow_forwardPractice Question 19. Consider the titration of 20.0 mL of 0.20 M NaOH with 0.1 M HCl, Calculate the initial pH and then the new PH after adding 10mL of HCLarrow_forward
- What is the pH of 1.00 L of the 0.100 M hydrofluoric acid–0.120 M fluoride ion buffer system described in Worked Example 10.16 after 0.020 mol of HNO3 is added?arrow_forwardHCN is a weak acid with a Ka = 4.90 • 10 . A 50.00 mL sample of this HCN solution (0.250 M) is titrated with 0.500 M NaOH. ______________(Answer) b) What is the pH after 15 mL of NaOH is added? ______________(Answer) c) What is the pH at the equivalence point? ______________(Answer) Rank the following in order of increasing pOH and justify: 0.10 M solutions of chloroacetic acid, carbonic acid, and citric acid Write out the order instead of placing 1, 2 ... Rank the following in order of increasing pKa and justify: 0.10 M mandelic acid, 0.20 M maleic acid, 0.40 M malonic acid. Write out the order instead of placing 1, 2 ...arrow_forwardQUESTION 24. Acetic acid is a convenient buffer system in the slightly acidic range. What mass of sodium acetate (NaCH3CO2) must be added to 0.750 L of 0.116 M solution of acetic acid to make a buffer of pH = 5.00? The Ka of acetic acid is 1.8E-5. How many moles of acid are required to change the pH of the solution in part a by 0.05 pH units?arrow_forward
- What is the change in pH with the addition of 0.01 mol HCl to a liter of a buffer solution containing 0.05M NH3 & 0.05M NH4Cl? The Kb value of NH3 is 1.80 x 10-5 at 25C. Answer in three decimal places Initial pH = ? pH with HCl = ? pH change = ?arrow_forwardA 27.3 mL sample of a 0.457 M aqueous acetic acid solution is titrated with a 0.447 M aqueous sodium hydroxide solution. What is the pH at the start of the titration, before any sodium hydroxide has been added?pH = Submit Answerarrow_forwardHow many milliliters of 0.100 M H3PO4 and 0.100 M NaOH solutions are required to prepare 1.0 L of a buffer of pH 6.90? (Ka1=7.5×10-3, Ka2=6.2×10-8, Ka3=4.5×10-13) (Answer: VH3PO4=429 mL, VNaOH=571 mL).arrow_forward
- Which of the following aqueous mixtures will result in a buffer with a pH higher than 7.0? (For HCNO, Ka = 2.2×10-4, for NH3, Kb = 1.8×10-5 ) 10 mL of 0.1 M NH3 + 10 mL of 0.1 M HCl 10 mL of 0.1 M NH3 + 10 mL of 0.1 M HCNO 10 mL of 0.1 M NH3 + 5.0 mL of 0.1 M HCl (Correct answer) 10 mL of 0.1 M HCNO + 10 mL of 0.1 M NaOH 10 mL of 0.1 M HCNO + 5.0 mL of 0.1 M NaOH please explain. thanks!arrow_forwardAn aqueous solution contains 0.414 M ethylamine (C2H5NH2).How many mL of 0.214 M nitric acid would have to be added to 225 mL of this solution in order to prepare a buffer with a pH of 10.500? Answer:_____arrow_forwardA beaker contains 34.141 mL of a 0.243 M solution of LiOH. This is titrated with 16.18 mL of a 0.0251 M solution of HClO3. What is the final pH? The solubility product constant for MX3 is 7.45E-18. What is the molar solubility of MX3? (SCIENTIFIC NOTATION) Which of the following acids is best used to prepare a buffer with a pH of 8.8? A. CH3COOH, pKa = 4.74 B. HBrO, pKa = 8.64 C. C5H5O5COOH, pKa = 5.40 D. C6H4(COOH)2, pKa = 3.54 E. HOC6H4OCOOH, pKa = 3.00 Fructose (molar mass = 180.2 g mol−1) is a common sugar. What is the molality of a solution of fructose when 22.4 grams of fructose have dissolved in enough water to make 323.5 mL of solution? The density of this final solution is 1.40 g mL−1. (the answer should be entered with 3 significant figures; do not enter units; give answer in normal notation; examples include 1.23 and 12.3 and -123 and 123.). NEED THESE ANSWERED ASAP NO WORK NEEDED THANSKarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
