
ORGANIC CHEMISTRY-PRINT (LL)-W/WILEY
4th Edition
ISBN: 9781119761105
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 4.2, Problem 2PTS
Interpretation Introduction
Interpretation:
The two compounds having the same parent chain should be identified among the structure of compounds mentioned below.
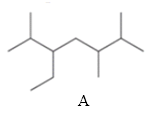
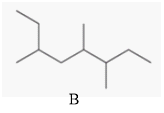
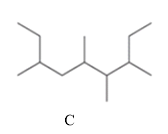
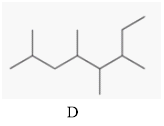
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Name the following structures
CH3
CH3 F
CH3
Br
H3C
H
CH3
H
CH3CH₂CH₂CH₂OH
Part A
What is the relationship between the following molecules?
CH,CH3
CH3
CH2
CHCH3
CH;CH2-CH- CH-CH,-CH-CH-CH3
CH3
CH,CH3
CH3
CHCH3
CH,CH3
CH;CH, CH-CH-CH,-CH–ĊH-CH,CH,CH3
CH3
CH3
O They are different molecules which are not isomers.
O They are identical.
O They are isomers of each other.
O none of the above
Submit
Request Answer
Identify the following.
Chapter 4 Solutions
ORGANIC CHEMISTRY-PRINT (LL)-W/WILEY
Ch. 4.2 - Prob. 1LTSCh. 4.2 - Prob. 1PTSCh. 4.2 - Prob. 2PTSCh. 4.2 - Prob. 3ATSCh. 4.2 - Prob. 2LTSCh. 4.2 - Prob. 4PTSCh. 4.2 - Prob. 5ATSCh. 4.2 - Prob. 3LTSCh. 4.2 - Prob. 6PTSCh. 4.2 - Prob. 7ATS
Ch. 4.2 - Prob. 4LTSCh. 4.2 - Prob. 8PTSCh. 4.2 - Prob. 9PTSCh. 4.2 - Prob. 10ATSCh. 4.2 - Prob. 5LTSCh. 4.3 - Prob. 6LTSCh. 4.3 - Prob. 14PTSCh. 4.6 - Prob. 7LTSCh. 4.6 - Prob. 16PTSCh. 4.8 - Prob. 8LTSCh. 4.12 - Prob. 11LTSCh. 4.12 - Prob. 25PTSCh. 4.14 - Prob. 33CCCh. 4.14 - Prob. 34CCCh. 4.14 - Prob. 35CCCh. 4 - Prob. 36PPCh. 4 - Prob. 51PPCh. 4 - Prob. 52PPCh. 4 - Prob. 53PPCh. 4 - Prob. 54PPCh. 4 - Prob. 55PPCh. 4 - Prob. 56PPCh. 4 - Prob. 57PPCh. 4 - Prob. 58PPCh. 4 - Prob. 59PPCh. 4 - Prob. 69ACPCh. 4 - Prob. 73IPCh. 4 - Prob. 76IP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structures of the following molecules: (a) trans-l-Bromo-3-methylcyclohexane (b) cis-1, 2-Dimethylcyclobutane (c) trans-1 -tert-Butyl-2-ethylcyclohexanearrow_forwardIdentify the carbon atom that should be designated as #1 when naming this compound systematically. CH3 Which carbon is C1? A E A -CH2CH3 H3C D E Give the systematic name for the compound. Spelling and punctuation count! Name:arrow_forwardExamples: Name the following compounds: Parent chain: Substituent(s): Position of each substituent: Name: Parent chain: Substituent(s): Position of each substituent: Name: Parent chain: Substituent(s): Position of each substituent: Name:arrow_forward
- Name the following organic compounds: CH3 I CH₂-CH-CH- compound - I CH₂ | CH3 CH3CH₂- -C- | CH3CH₂- CH- CH3 I CH₂- - CH₂ - CH₂ - CH₂ - CH3 CH₂ - CH₂ - CH3 CH3 CH₂-CH- CH- I CH3 CH₂ - CH₂ name A U X Sarrow_forwardProvide a systematic name of the following compound below: sec-pentyl cyclohexane ہےarrow_forwardQ)Name the following heterocyclic compounds CH3 Br M euwe CH3arrow_forward
- Name the following compounds: a Br CH-C=C-C-CHる CH3 d. Br e.arrow_forwardClassify the following compounds as E/Z- structures CH3 HO-CH2-CH2 CH2CHCH2OH H2N CH2-CH3 H2N CH=CH2 CH2=CH I CH-CH3 но- CH II CH2-NH2 Br CH2CH2CH2CH3 III CH3 OHarrow_forwardDraw the structures of the following heterocyclic compounds:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
