
ORGANIC CHEMISTRY
4th Edition
ISBN: 9781119745105
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 4.2, Problem 5ATS
Interpretation Introduction
Interpretation:
The identification and name of all the substituents in the following compound should be determined.
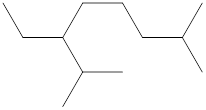
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
3-Methyl-2-hexenoic acid (mixture of E and Z isomers) has been identified as the substance responsible for the odor of human sweat. Synthesize the compound from raw materials that have five carbons or less.
Both 1,2-dihydronaphthalene and 1,4-dihydronaphthalene may be selectively hydrogenated to 1,2,3,4-tetrahydronaphthalene.One of these isomers has a heat of hydrogenation of 101 kJ/mol (24.1 kcal/mol), and the heat of hydrogenation of the other is 113 kJ/mol (27.1 kcal/mol). Match the heat of hydrogenation with the appropriate dihydronaphthalene.
When 2-chloropropane treated with NaOH and 1-chloropropane treated with NaOH separately produce two different functional groups. Provide both reactions and explain the two different functional groups produced.
Chapter 4 Solutions
ORGANIC CHEMISTRY
Ch. 4.2 - Prob. 1LTSCh. 4.2 - Prob. 1PTSCh. 4.2 - Prob. 2PTSCh. 4.2 - Prob. 3ATSCh. 4.2 - Prob. 2LTSCh. 4.2 - Prob. 4PTSCh. 4.2 - Prob. 5ATSCh. 4.2 - Prob. 3LTSCh. 4.2 - Prob. 6PTSCh. 4.2 - Prob. 7ATS
Ch. 4.2 - Prob. 4LTSCh. 4.2 - Prob. 8PTSCh. 4.2 - Prob. 9PTSCh. 4.2 - Prob. 10ATSCh. 4.2 - Prob. 5LTSCh. 4.3 - Prob. 6LTSCh. 4.3 - Prob. 14PTSCh. 4.6 - Prob. 7LTSCh. 4.6 - Prob. 16PTSCh. 4.8 - Prob. 8LTSCh. 4.12 - Prob. 11LTSCh. 4.12 - Prob. 25PTSCh. 4.14 - Prob. 33CCCh. 4.14 - Prob. 34CCCh. 4.14 - Prob. 35CCCh. 4 - Prob. 36PPCh. 4 - Prob. 51PPCh. 4 - Prob. 52PPCh. 4 - Prob. 53PPCh. 4 - Prob. 54PPCh. 4 - Prob. 55PPCh. 4 - Prob. 56PPCh. 4 - Prob. 57PPCh. 4 - Prob. 58PPCh. 4 - Prob. 59PPCh. 4 - Prob. 69ACPCh. 4 - Prob. 73IPCh. 4 - Prob. 76IP
Knowledge Booster
Similar questions
- Give IUPAC names for the following compounds:arrow_forwardMethyl isocyanate, CH3 -N= C = O, is used in the industrial synthesis of a type of pesticide and herbicide known as a carbamate. As a historical note, an industrial accident in Bhopal, India, in 1984 resulted in leakage of an unknown quantity of this chemical into the air. An estimated 200,000 people were exposed to its vapors, and over 2000 of these people died. Q.) Methyl isocyanate reacts with strong acids, such as sulfuric acid, to form a cation. Will this molecule undergo protonation more readily on its oxygen or nitrogen atom? In considering contributing structures to each hybrid, do not consider structures in which more than one atom has an incomplete octetarrow_forwardThree products with the molecular formula C6 H4BrCl form when bromobenzene is treated with chlorine, Cl2, in the presence of FeCl3 as a catalyst. Name and draw a structural formula for each product.arrow_forward
- 2. The molecules shown below are four terpineol isomers. They are isolated from plants and have distinct aromas. For example, a -terpineol is a common perfume ingredient and smells similar to lilacs. Hol H-O- a-terpineol B-terpineol Y-terpineol 4-terpineol A) Draw a structural isomer of terpineol that meet the following criteria. You should draw one unique structure for each set. You must draw each molecule twice, once as a skeletal structure and once as a complete Lewis structure showing all atoms, bonds, and lone pairs of electrons. i. Does not have a ring. ii. Has an E alkene. iii. Has a secondary alcohol. iv. Does not contain an alcohol. B) Draw a complete Lewis structure of a-terpineol showing all bonds, atoms and lone pairs of electrons.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardCan an aldehyde have molecular formula C 5H 12O? Explain why or why not.arrow_forward2) Provide an acceptable name for the compound below. OH CH₂CH₂CH3 106 gaidie than methanol Use appropriatearrow_forward
- For each of the following, draw one structural formula that meets all stated requirements, then name each structure you drew according to either IUPAC or common nomenclature.arrow_forwardExplain why methyl trifluoroacetate, CF3CO2CH3, is more reactive than methyl acetate, CH3CO2CH3, in nucleophilic acyl substitution reactions.arrow_forwardCitronellol ((3R)-3,7-dimethyloct-6-en-1-ol, C10H20O) is an organic fragrance found in the oil extracted from lemon grass. a) Name the two functional groups present in the molecule. b) When a few drops of bromine dissolved in hexane is added to a sample of citronellol the brown colour of the bromine rapidly disappears. i. What type of chemical reaction has occurred? ii. Draw the structure of the product formed. iii. Name the product. c) The product formed when citronellol is heated with a mixture of potassium dichromate and sulfuric acid gives a yellow/orange precipitate when shaken with Brady’s reagent (2,4-dinitrophenylhydrazine). It also shows a positive result with Fehling’s solution. i. What type of chemical reaction has occurred ? ii. What type of functional group is present in the product? d) Explain the type of stereoisomerism which may occur in citronellol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
