
Concept explainers
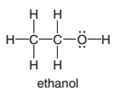
(a)
Interpretation:
The shape around the carbon in ethanol along with it is polar or non-polar is to be determined.
Concept Introduction:
The polar bond is formed between the two unlike atoms having electronegative difference ranges from 0.5-1.9. The polar bond represents the arrow towards the high electronegative element, while the tail towards the low electronegative element. The negative sign is given to the high electronegative element while a positive sign is given to the low electronegative element.
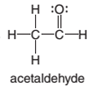
(b)
Interpretation:
The shape around the carbon in acetaldehyde along with it is polar or non-polar is to be determined.
Concept Introduction:
The polar bond is formed between the two unlike atoms having electronegative difference ranges from 0.5-1.9. The polar bond represents the arrow towards the high electronegative element, while the tail towards the low electronegative element. The negative sign is given to the high electronegative element while a positive sign is given to the low electronegative element.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, & Biological Chemistry
- Draw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH3CH2)2O, the rst general anesthetic used in medical proceduresb. acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon bersc. dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning productsd. acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forwardWhat is the difference between the hybridization of carbon atoms' valence orbitals in saturated and unsaturated hydrocarbons?arrow_forwardClassify each pair of compounds as isomers or resonance structures ?arrow_forward
- Unlike methanol, which is a nearly odorless liquid, methanethiol (CH3SH) is a gas with an appalling odor reminiscent of skunks. It is one of the compounds added to natural gas as a warning for gas leaks. Using appropriate drawing tools, draw the structure of methanethiol in such a way that there is double bond between carbon and sulfur.arrow_forwardWhat molecule(s) are primarily responsible for the bitter taste of dark chocolate (the darkest chocolate is synonymous with bittersweet)?arrow_forwardConvert each condensed formula to a Lewis structure. a.CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c. (CH3)2CHCHO d.(HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forward
- Each carbon molecule can bond with as many as________ other atom(s) or molecule(s). a. one b. two c. six d. fourarrow_forwardConvert each condensed formula to a Lewis structure. CH3(CH2)4CH(CH3)2 (CH3)3CCH(OH)CH2CH3 (CH3)2CHCHO (HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardWhich molecules show an appropriate number of bonds around each carbon atom?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
