
EBK ORGANIC CHEMISTRY
12th Edition
ISBN: 9781119233664
Author: Snyder
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 13PP
Practice Problem 5.13
Tell whether the two structures in each pair represent enantiomers or two molecules of the same compound in different orientations.
(a)

(b)
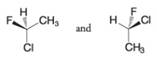
(c)
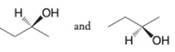
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
5.76 2,3-Dibromoprop-1-ene (C3H4Br₂) has four H atoms. Suppose that any of these H atoms can be replaced by a Cl atom to
yield a molecule with the formula C3H3Br₂Cl. (a) Identify two H atoms where this substitution would yield constitutional
isomers of C3H3Br₂Cl; (b) enantiomers of C3H3Br₂Cl; (c) diastereomers of C3H3Br₂Cl.
co
11
HHH
Juods H Brugtrio 2 bas A
2,3-Dil
so ontemme A
chon
Br
2,3-Dibromoprop-1-ene
(B) Draw two chair conformations of the following substituted cyclohexane and identify the
most stable conformation. Clearly show the axial and equatorial bonds.
2.35
Write a structural formula for each of the following compounds:
(a) 6-Isopropyl-2,3-dimethylnonane
(b) 4-tert-Butyl-3-methylheptane
(c) 4-Isobutyl-1,1-dimethylcyclohexane
(d) sec-Butylcycloheptane
(e) Cyclobutylcyclopentane
Chapter 5 Solutions
EBK ORGANIC CHEMISTRY
Ch. 5 - Prob. 1PPCh. 5 - Prob. 2PPCh. 5 - Prob. 3PPCh. 5 - Prob. 4PPCh. 5 - Prob. 5PPCh. 5 - Prob. 6PPCh. 5 - Prob. 7PPCh. 5 - Practice Problem 5.8 Write three-dimensional...Ch. 5 - Prob. 9PPCh. 5 - Prob. 10PP
Ch. 5 - Practice Problem 5.11 List the substituents in...Ch. 5 - Prob. 12PPCh. 5 - Practice Problem 5.13 Tell whether the two...Ch. 5 - Prob. 14PPCh. 5 - Prob. 15PPCh. 5 - Prob. 16PPCh. 5 - Prob. 17PPCh. 5 - Prob. 18PPCh. 5 - Prob. 19PPCh. 5 - Prob. 20PPCh. 5 - Practice Problem 5.21 The following are formulas...Ch. 5 - Practice Problem 5.22 Write three-dimensional...Ch. 5 - Prob. 23PPCh. 5 - Practice Problem 5.24 Give names chat include (R)...Ch. 5 - Prob. 25PPCh. 5 - Prob. 26PPCh. 5 - Prob. 27PPCh. 5 - Prob. 28PPCh. 5 - Practice Problem 5.29 Write formulas for all of...Ch. 5 - Prob. 30PPCh. 5 - Prob. 31PPCh. 5 - Prob. 32PPCh. 5 - Prob. 33PCh. 5 - Prob. 34PCh. 5 - 5.35 Designate the (R) or (S) configuration at...Ch. 5 - Prob. 36PCh. 5 - (a) Write the structure of...Ch. 5 - Shown below are Newman projection formulas for...Ch. 5 - 5.39 Write appropriate structural formulas...Ch. 5 - Discuss whether each of the following compounds...Ch. 5 - Prob. 42PCh. 5 - Prob. 43PCh. 5 - Compound F has the molecular formula C5H8 and is...Ch. 5 - Prob. 45PCh. 5 - Prob. 46PCh. 5 - Prob. 47PCh. 5 - For the following molecule, draw its enantiomer as...Ch. 5 - 5.49 (Use models to solve this...Ch. 5 - 5.50 (Use models to solve this...Ch. 5 - (Use models co solve this problem.) Write...Ch. 5 - 5.52 Tartaric acid was an important compound in...Ch. 5 - Prob. 53PCh. 5 - Prob. 54PCh. 5 - Prob. 55PCh. 5 - Prob. 1LGPCh. 5 - Prob. 2LGPCh. 5 - Prob. 3LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Thiols such as ethanethiol and propanethiol can be used to reduce vitamin K epoxide to vitamin KH2, but they re...
Organic Chemistry
Practice Exercise 2
By using a conversion factor from the back inside cover, determine the length in kilometer...
Chemistry: The Central Science (13th Edition)
2. A schedule of experiments for a laboratory indicates that the 1-bromobutane preparation is paired with the p...
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
A compound that contains only C and H was burned in excess O2 to give CO2 and H2O. When 0.270 g of the compound...
General Chemistry: Atoms First
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2.35 Consider 1-bromo-2-methylpropane and draw the following.(a) The staggered conformation(s) of lowest energy(b) The staggered conformation(s) of highest energyarrow_forwardConsider 1-bromopropane, CH3CH2CH2Br. (a) Draw a Newman projection for the conformation in which CH3 and -Br are anti (dihedral angle 180°). (b) Draw Newman projections for the conformations in which - CH3 and -Br are gauche (dihedral angles 60° and 300°). (c) Which of these is the lowest energy conformation? (d) Which of these conformations, if any, are related by reflection?arrow_forwardFor each of the following molecular formulas, determine the number of elements of unsaturation,and draw three examples.(a) C4H4Cl2 (b) C4H8Oarrow_forward
- 2.34 Consider 1-bromopropane, CH3CH2CH2Br.(a) Draw a Newman projection for the conformation in which !CH3 and !Br are anti (dihedral angle 1808).(b) Draw Newman projections for the conformations in which !CH3 and !Br are gauche (dihedral angles 608 and 3008).(c) Which of these is the lowest energy conformation?(d) Which of these conformations, if any, are related by reflection?arrow_forward(a) Draw a skeletal structure of the anabolic steroid methenolone from the following description. Methenolone contains the tetracyclic steroid skeleton with a carbonyl group at C3, a hydroxyl at C17, a double bond between C1 and C2, and methyl groups bonded to C1, C10, and C13. (b) Add wedges and dashed wedges for all stereogenic centers with thefollowing information: the configuration at C10 is R, the configuration at C13 is S, the configuration at C17 is S, and all substituents at ring fusions are trans to each other. (c) Draw the structure of Primobolan, the product formed when methenolone is treated with CH3(CH2)5COCl and pyridine. Primobolan is an anabolic steroid that can be taken orally or by injection and has been used illegally by well-known Major League Baseball players.arrow_forwardDetermine whether the following pairs of structures in each set represent the same molecule or constitutional isomers, and if they are the same molecule, determine whether they are in the same or different conformations: (a) (b) (c) (d)arrow_forward
- 3B. For each compound below, draw two chair conformations. Indicate whether the substituents are axial or equatorial. Indicate which chair conformation is more stable. (you need to draw conformations of the exact molecule, not its mirror image or diastereomer) (a) (b) (c) Ме Me Et "Et Et Mearrow_forward(a) which if the structure of trans-1,2-dimethylcyclopentane? (b) which is the most stable conformation of 1-bromo-2-ethylcyclohexane? (c) which is the least stable conformation of 1-bromo-2-ethylcyclohexane? (d) which is the more stable configuration of 1,3-dimethylcyclopentane? *Et = ethylarrow_forwardThe cis ketone A is isomerized to a trans ketone B with aqueous NaOH. Asimilar isomerization does not occur with ketone C. (a) Draw thestructure of B using a chair cyclohexane. (b) Label the substituents in Cas cis or trans, and explain the difference in reactivity.arrow_forward
- (a) Convert each chair cyclohexane to a hexagon with wedges and dashed wedges. (b) Draw a stereoisomer of each compound in its more stable chair conformation.arrow_forwardTake a look at the butane conformers below. Identify: (a)Which is an anti conformation in Newman? (b)Which is a Gauche conformation? (c)Which is the more stable Sawhorse conformer? (d)Which has the same potential energy/strain with ALS?arrow_forward(a) Write the structures of the following compounds and mark them as chiral or achiral. 4 (i) 2-Bromopentane (ii) 3-Bromopentane (iii) 1-Bromo-2-methylbutane (iv) 2-Chloro-3-methylbutane (b) Identify the asymmetric carbon in the chiral compounds. (c) Write the structure of the other enantiomer of the chiral compounds.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License