
SCIENTIFIC THINKING Mercury is known to inhibit the permeability of water channels. To help establish that the protein isolated by Agre’s group was a water channel (see Module 5.7), the researchers incubated groups of RNA-injected oocytes (which thus made aquaporin proteins) in four different solutions: plain buffer, low concentration and high concentration of a mercury chloride (HgCl2) solution, and low concentration of a mercury solution followed by an agent (ME) known to reverse the effects of mercury. The water permeability of the cells was determined by the rate of their osmotic swelling. Interpret the results of this experiment, which are presented in the graph below.
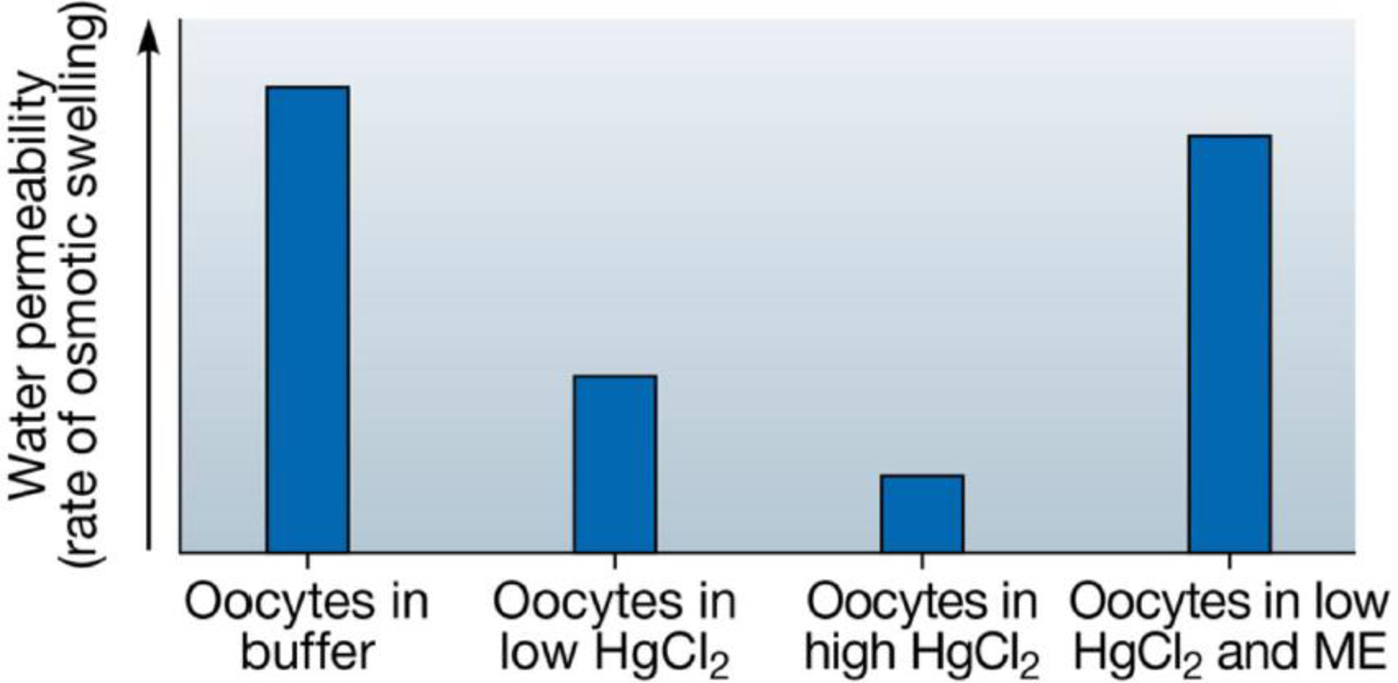
Data from G. M. Preston et al., Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein, Science 256: 3385–7 (1992).
Control oocytes not injected with aquaporin RNA were also incubated with buffer and the two concentrations of mercury. Predict what the results of these treatments would be.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
CAMPBELL BIO&VALPK A/C MAST PKG
- A. Assumed that the primary contributor to the absorbance of the samplewas due to the hemoglobins. Calculate how much red light the water in the sample absorbed.Explain whether or not ignoring water was a reasonable assumption. (use image down below to solve) B. There are several membrane-bound organelles in cells (e.g. nuclei, mitochondria,endoplasmic reticulum) that can also have ion channels in their membranes. Sketch anelectrical circuit model of a cell that expresses sodium, calcium, and chloride ion channels in itscellular membrane and calcium and chloride ion channels in its nuclear envelope membrane.arrow_forwardPart a) Would a single alpha helix be more stable in a aqueous solution or a membrane. Explain why. Part b) Why do transmembrane proteins always have secondary structures inside the membrane but aqueous proteins do not? Part c) If you had a single alpha helix, can it act as a pore for a membrane. Explain.arrow_forwardFor osmosis in science, if you gargle with salt water why does your throat stop hurting?arrow_forward
- For a lab on diffusion and osmosis, where a egg yolk was placed in a cup water the following question asks Consider a scenario in which the size of an egg yolk remains unchanged after in water soaking for an hour. What are two possible explanations as to why this occurs? What would two reasons be for the yolk to be unchanged after a hour in water?arrow_forwardCold-blooded organisms may adapt to changing environments by making their membranes more fluid when it is very cold or less fluid when it is very hot heat. Explain in 20 words or fewer how an organism's membranes would change at the molecular level to adapt to a very hot environmentarrow_forwardIsotonic saline and 5% dextrose in water are solutions considered isotonic to human blood. What effect on red blood cells would you expect if a patient were given these fluids intravenously? A solution of 10% dextrose in water is hypertonic to human blood.arrow_forward
- Biology Cell adhesion can often be blocked in vitro by treating cells with specific agents. Which of the enzymes (out of Trypsin; neuraminidase; collagenase; hyaluronidase) would be expected to interfere with cell adhesion mediated by: a. selectins? b. with cell adhesion mediated by L1 molecules? Also explain the underlying mechanism of this interferencearrow_forward1. It can separate large colloidal particles from small solute particles of a solution. a. Dialysis b. Facilitated diffusion c. Osmosis d. Diffusion 2. What force of interaction was cleaved in the addition of a reducing agent to the protein sample? a. Disulfide bonds b. hydrogen bond c. hydrophobic interactions d. ionic bond 3. The substance in the vinegar that dissolved the egg shell. a. calcium acetate b. calcium carbonate c. water d. acetic acid 4. As enzyme concentration increases, enzymatic activity increases provided the substrate concentration is constant. True or False 5. Diffusion is the separation of smaller molecules from larger molecules using a semi-permeable membrane. True or Falsearrow_forwardYou are a doctor in the emergency room. You have a patient whose blood cells are composed of the following: 80 molecules of glucose, 50 molecules of hemoglobin, 20 molecules of dextrose, and 2000 molecules of water. a. What is the total solute concentration of her cells? b. If you needed to make an isotonic solution of saline for her IV, how many salt molecules would you need to add to 1000 molecules of water? How do you know this is the correctamount?arrow_forward
- A concentration gradient for water must be present in cells for osmosis to occur. Which bag represented the steepest concentration gradient relative to its surrounding environment?arrow_forwardyeasts are able to produce high internal concentrations of glycerol to counteract the osmotic pressure of the surrounding media. suppose that a sample of yeast cells were placed in a 4% sodium chloride solution by weight. The density of solution is at 25 C = 1.02 g/ml, Molecular weight of solute = 58.44 g/mol, i of glycerol = 1 and R=0.08205 L-atm/mol-K What is the weight of solute in grams What is the moles of solute What is the volume of the solution in liters What is the molarity of the solution What is the value of the temperature to be used to solved for the osmotic pressure of the solution What is the osmotic pressure of solutionarrow_forwardNa+ moves from high to low concentration, across the plasma membrane, through a protein channel that is permenantly open. Which statement is true about this process? a. It does not require ATP b. It is a form of active transport c. Movement is against the concentration gradient of Na+ d. The channel probably allows many different molecules and ions to crossarrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





