
Concept explainers
(a)
Interpretation:
Atomic orbitals which are used to form each
Concept Introduction:
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Sigma (σ) bonds are the bonds in which shared hybrid orbital’s electron density are concentrated along the internuclear axis.
Pi (π) bonds are the bonds in which shared unhybridized orbital’s (p, d, etc) electron density are concentrated in above and below of the plane of the molecule.
Geometry of different types of molecule with respect to the hybridizations are mentioned are mentioned below,
(a)
Explanation of Solution
In the marked carbon atom, one s and three p orbital hybridize forming four
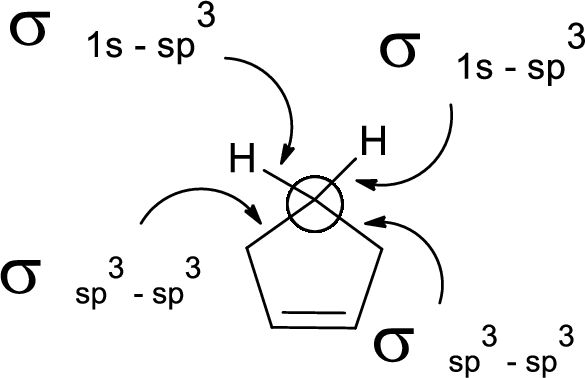
In the marked carbon atom, one s and two p orbital hybridize forming three
(b)
Interpretation:
Atomic orbitals which are used to form each
Concept Introduction:
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Sigma (σ) bonds are the bonds in which shared hybrid orbital’s electron density are concentrated along the internuclear axis.
Pi (π) bonds are the bonds in which shared unhybridized orbital’s (p, d, etc) electron density are concentrated in above and below of the plane of the molecule.
Geometry of different types of molecule with respect to the hybridizations are mentioned are mentioned below,
(b)
Explanation of Solution
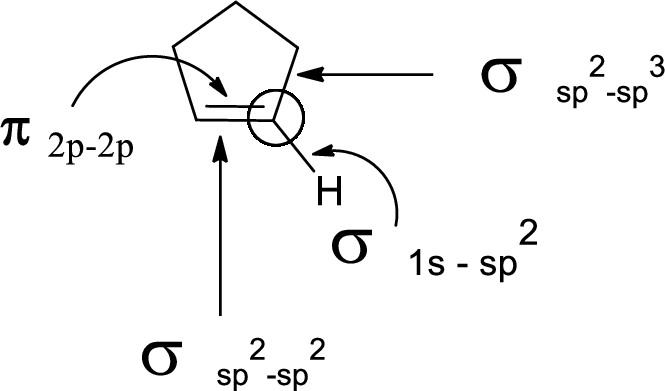
In the marked carbon atom, one s and two p orbital hybridize forming three
(c)
Interpretation:
Atomic orbitals which are used to form each
Concept Introduction:
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Sigma (σ) bonds are the bonds in which shared hybrid orbital’s electron density are concentrated along the internuclear axis.
Pi (π) bonds are the bonds in which shared unhybridized orbital’s (p, d, etc) electron density are concentrated in above and below of the plane of the molecule.
Geometry of different types of molecule with respect to the hybridizations are mentioned are mentioned below,
(c)
Explanation of Solution
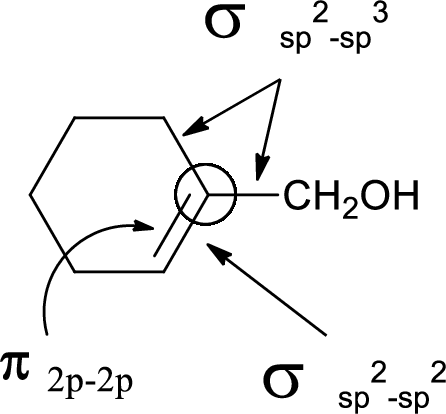
In the marked carbon atom, one s and two p orbital hybridize forming three
(d)
Interpretation:
Atomic orbitals which are used to form each
Concept Introduction:
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Sigma (σ) bonds are the bonds in which shared hybrid orbital’s electron density are concentrated along the internuclear axis.
Pi (π) bonds are the bonds in which shared unhybridized orbital’s (p, d, etc) electron density are concentrated in above and below of the plane of the molecule.
Geometry of different types of molecule with respect to the hybridizations are mentioned are mentioned below,
(d)
Explanation of Solution
In the marked carbon atom, one s and two p orbital hybridize forming three
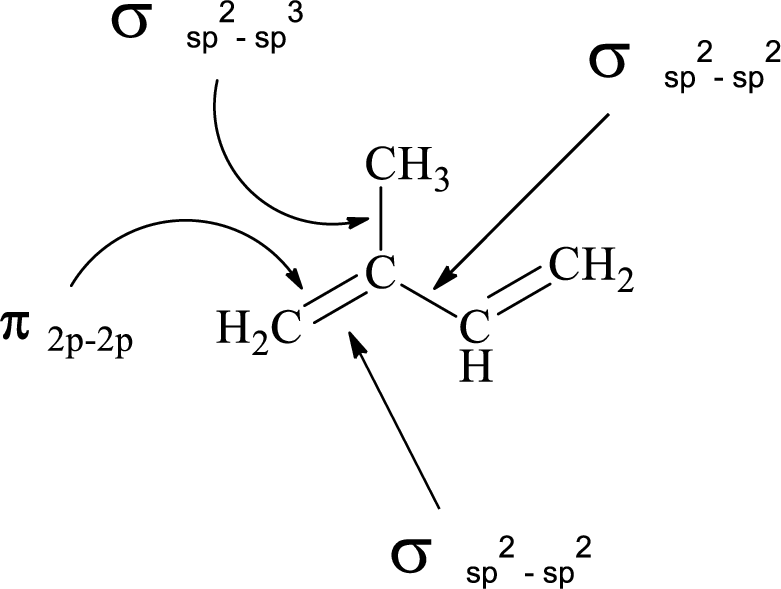
(e)
Interpretation:
Atomic orbitals which are used to form each
Concept Introduction:
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Sigma (σ) bonds are the bonds in which shared hybrid orbital’s electron density are concentrated along the internuclear axis.
Pi (π) bonds are the bonds in which shared unhybridized orbital’s (p, d, etc) electron density are concentrated in above and below of the plane of the molecule.
Geometry of different types of molecule with respect to the hybridizations are mentioned are mentioned below,
(e)
Explanation of Solution
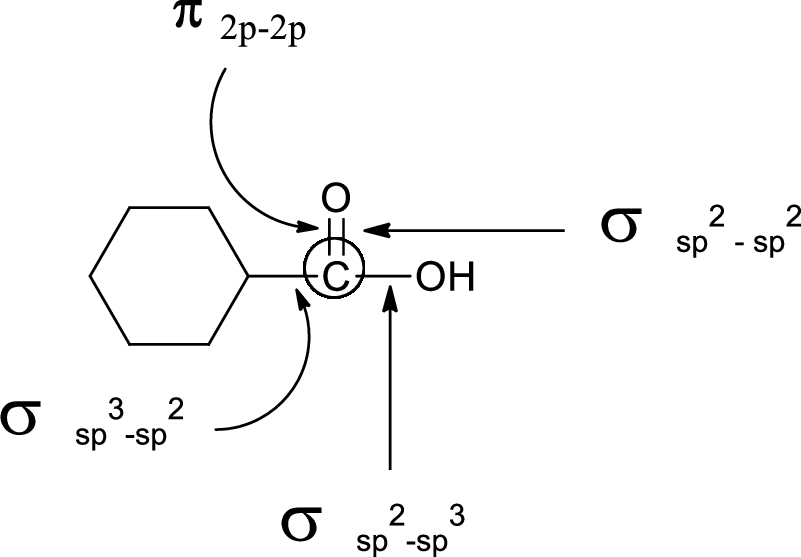
In the marked carbon atom, one s and one p orbital hybridize forming two
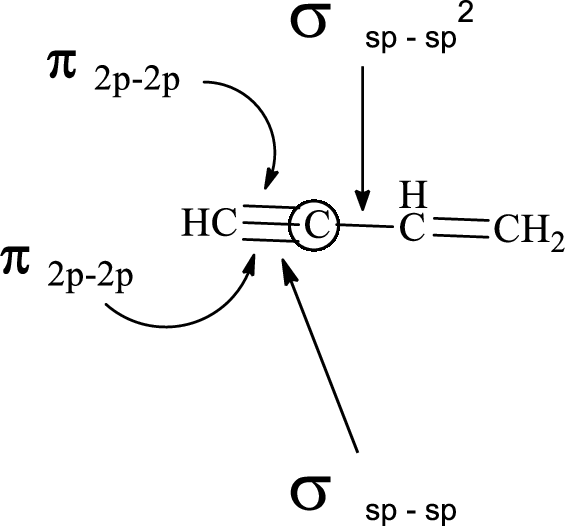
In the marked carbon atom, one s and two p orbital hybridize forming three
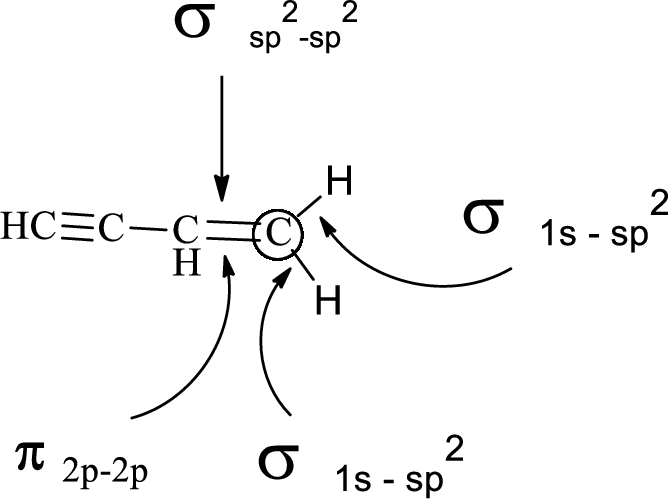
Want to see more full solutions like this?
Chapter 5 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- 1. Explain the two ways that methane (CH4) illustrates the need for hybridized molecular bonds. Draw a picture to represent the hybridization of carbon's orbitals and their overlap with hydrogen's. Label each orbital used in bonding with its letters and each bond as sigma or pi.arrow_forwardWhen looking at the hybridized orbital diagram, we can see which orbitals( and their electrons) will lead to sigma, pi and lone pair of electrons. Indicate where you would find each of the following :arrow_forwardplease help a bit confusedarrow_forward
- Draw a three-dimensional representation of the valence orbitals and their bonding for compound CH2CHOH. Clearly show all valence electrons for each atom. Provide the following labels: atomic symbols, a code for each type of orbital used, a sigma bond, a pi bond and the molecular shape and approximate bond angles around the non-hydrogen atoms.arrow_forward3) How many electrons are delocalized in how many p-orbitals? Draw the most and second most stable resonance structures of the following compound. Order the structures by increasing stability. What is the partial charge distribution in the resonance hybrid?arrow_forwardHow does the valence bond description of a carboncarbon double bond account for cis trans isomers?arrow_forward
- This is the shape of the sp hybrid carbons in this compound. tetrahedral trigonal planar linear square This is the General Formula of alkanes, where n is the number of C. CnH2n CnH2n-2 CnH2n+2 CnH2n-4 This class of organic compounds is saturated with only carbon-carbon single bonds. O Alkynes Alkenes Alkanes Arenes O Oarrow_forwardThese molecules are called constitutional (or structural) isomers because they share the same chemical formula but have different patterns of atomic connectivity. Draw as many constitutional isomers as you can for C5H8 in bond-line notation. It may be helpful to draw a Lewis Structure for one of the isomers first.arrow_forward8. a) State the hybridization (sp°, sp', sp) of each carbon in this molecule, going left to right. HC b) Write "most" under the most stable alkene. Write "least" under the least stable alkene. c) Write "most" under the most stable cation. Write "least" under the least stable cation. CH2 CH3 CH3 CH3 CH3 H3C CHT HC H3C ONLY roactions we have studied this semester.arrow_forward
- : The three bonds in the carbon-carbon triple bond in H-C≡C-H are:a _______ bond formed from _______ orbitalsa _______ bond formed from _______ orbitalsa _______ bond formed from _______ orbitalsarrow_forwardMethyl isocyanate, CH3 -N= C = O, is used in the industrial synthesis of a type of pesticide and herbicide known as a carbamate. As a historical note, an industrial accident in Bhopal, India, in 1984 resulted in leakage of an unknown quantity of this chemical into the air. An estimated 200,000 people were exposed to its vapors, and over 2000 of these people died. Q.) Write a Lewis structure for methyl isocyanate and predict its bond angles. What is the hybridization of its carbonyl carbon? Of its nitrogen atom?arrow_forwardFor highlighted carbon atom , identify which atomic orbitals are used to form each σ bond and which are used to form each π bond.arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


