![OWLv2 for Ebbing/Gammon's General Chemistry, 11th Edition, [Instant Access], 1 term (6 months)](https://s3.amazonaws.com/compass-isbn-assets/textbook_empty_images/large_textbook_empty.svg)
Concept explainers
The graph here represents the distribution of molecular speeds of hydrogen and neon at 200 K.
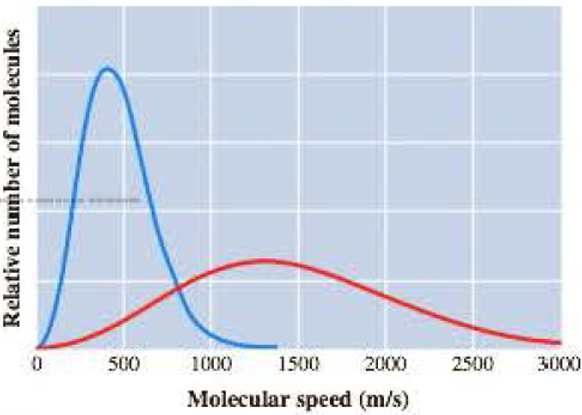
- a Match each curve to the appropriate gas.
- b Calculate the rms speed (in m/s) for each of the gases at 200 K.
- c Which of the gases would you expect to have the greater effusion rate at 200 K? Justify your answer.
- d Calculate the temperature at which the rms speed of the hydrogen gas would equal the rms speed of the neon at 200 K.
(a)
Interpretation:
The curve in the given graph must be matched with the appropriate gas.
Answer to Problem 5.136QP
The taller and narrow curve represents neon atoms
The flatter and wider curve represents hydrogen molecules
Explanation of Solution
Given,
The given graph represents the distribution of molecular speeds of hydrogen and neon at
Matching of given curves with appropriate gases:
In the given graph, the taller and narrow curve whose maximum falls near
The flatter and wider curve whose maximum falls near
The taller and narrow curve matches with neon atoms
The flatter and wider curve matches with hydrogen molecules
(b)
Interpretation:
The rms speed (in
Concept Introduction:
Root-mean-square (rms):
The root-mean-square molecular speed (
Where,
Answer to Problem 5.136QP
The rms speed of neon gas at
The rms speed of hydrogen gas at
Explanation of Solution
Given,
The given graph represents the distribution of molecular speeds of hydrogen and neon at
Calculation of rms speed:
The rms speed of neon is calculated as follows,
The rms speed of hydrogen gas is calculated as follows,
The rms speed of neon gas at
The rms speed of hydrogen gas at
(c)
Interpretation:
The gas that has greater effusion rate at
Concept Introduction:
Graham’s law of effusion:
Answer to Problem 5.136QP
The gas that has greater effusion rate at
Explanation of Solution
Given,
The given graph represents the distribution of molecular speeds of hydrogen and neon at
Gas possessing greater effusion rate:
The rates of effusion are directly related to the rms speed.
Greater the rms speed, greater is the rate of effusion.
Since the rms speed of hydrogen is greater than neon, hydrogen gas will have greater effusion rate.
In the container, the fast moving molecules collide with the holes of the container more often and hence have a higher effusion probability.
The gas that has greater effusion rate at
(d)
Interpretation:
The temperature at which the rms speed of the hydrogen gas equals the rms speed of neon gas at
Concept Introduction:
Root-mean-square (rms):
The root-mean-square molecular speed (
Where,
Answer to Problem 5.136QP
The temperature at which the rms speed of the hydrogen gas equals the rms speed of neon gas at 200 K is
Explanation of Solution
Given,
The given graph represents the distribution of molecular speeds of hydrogen and neon at
Temperature calculation:
The temperature equaling the rms speed of neon is calculated from root mean square equation as follows,
The temperature at which the rms speed of the hydrogen gas equals the rms speed of neon gas at
Want to see more full solutions like this?
Chapter 5 Solutions
OWLv2 for Ebbing/Gammon's General Chemistry, 11th Edition, [Instant Access], 1 term (6 months)
Additional Science Textbook Solutions
Fundamentals of Heat and Mass Transfer
Chemistry: A Molecular Approach
Chemistry by OpenStax (2015-05-04)
Chemistry: The Molecular Nature of Matter
CHEMISTRY-TEXT
Elementary Principles of Chemical Processes, Binder Ready Version
- Two identical He-filled balloons, each with a volume of 20 L, are allowed to rise into the atmosphere. One rises to an altitude of 3000 m while the other rises to 6000 m. a Assuming that the balloons are at the same temperature, which balloon has the greater volume? b What information would you need in order to calculate the volume of each of the balloons at their respective heights?arrow_forwardIf 4.83 mL of an unknown gas effuses through a hole in a plate in the same time it takes 9.23 mL of argon, Ar, to effuse through the same hole under the same conditions, what is the molecular weight of the unknown gas?arrow_forwardShown below are three containers of an ideal gas (A, B, and C), each equipped with a movable piston (assume that atmospheric pressure is 1.0 atm). a How do the pressures in these containers compare? b Are all the gases at the same temperature? If not, compare the temperatures. c If you cooled each of the containers in an ice-water bath to 0.0C, describe how the volumes and pressures of the gases in these containers would compare.arrow_forward
- A mixture of chromium and zinc weighing 0.362 g was reacted with an excess of hydrochloric acid. After all the metals in the mixture reacted, 225 mL dry of hydrogen gas was collected at 27C and 750. torr. Determine the mass percent of Zn in the metal sample. [Zinc reacts with hydrochloric acid to produce zinc chloride and hydrogen gas; chromium reacts with hydrochloric acid to produce chromium(III) chloride and hydrogen gas.]arrow_forwardA 19.9-mL volume of a hydrochloric acid solution reacts completely with a solid sample of magnesium carbonate, producing 183 mL of CO2 that is collected over water at 24.0C and 738 torr total pressure. The reaction is 2HCl(aq)+MgCO3(s)CO2(g)+H2O(l)+MgCl2(aq) What is the molarity of the HCl solution?arrow_forward99 Pure gaseous nitrogen dioxide (NO2) cannot be obtained, because NO2dimerizes, or combines with itself, to produce a mixture of NO2 and N2O4. A particular mixture of NO2, and N2O4 has a density of 2.39 g/L at 50°C and 745 torr. What is the partial pressure of NO2 in this mixture?arrow_forward
- Workers at a research station in the Antarctic collected a sample of air to test for airborne pollutants. They collected the sample in a 1.00-L container at 764 torr and 20 C. Calculate the pressure in the container when it was opened for analysis in a particulate-free clean room in a laboratory in South Carolina, at a temperature of 22 C.arrow_forwardStarting with the definition of rate of effusion and Graham’s finding relating rate and molar mass, show how to derive the Graham’s law equation, relating the relative rates of effusion for two gases to their molecular masses.arrow_forwardA mixture at 33 °C contains H2at 325 torr. N;at 475 tore and O2at 650. torr. What is the total pressure of the gases in the system? Which gas contains the greatest number of moles?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





