
BNDL: ACP ORGANIC CHEMISTRY:CH EM 231(W/ACCESS CARD)
8th Edition
ISBN: 9781337687539
Author: Brown/Iverson/Anslyn/ Foote
Publisher: CENGAGE C
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 5, Problem 5.29P
Interpretation Introduction
Interpretation:
Structural formula of vitamin A has to be drawn showing the four isoprene units linked by head-to-tail and cross-linked at one point forming vitamin A.
Concept Introduction:
Terpenes are made by joining five-carbon units, usually in a head to tail-fashion.
Isoprene unit:
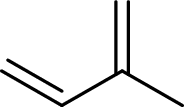
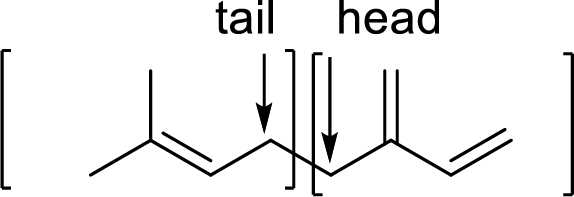
Branched end of isoprene – Head
Unbranched end of isoprene - Tail
Cross-link is a type of bond that links
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
DHA is a fatty acid derived from sh oil and an abundant fatty acid in vertebrate brains. Hydrogenation of DHA forms docosanoic acid [CH3(CH2)20CO2H] and ozonolysis forms CH3CH2CHO, CH2(CHO)2 (ve equivalents), and HCOCH2CH2CO2H. What is the structure of DHA if all double bonds have the Z conguration?
In addition to more highly fluorinated products, fluorination of 2-methylbutane yields a mixture of compounds with the formula C5H10F2. Draw the structures of all the isomers with the formula C5H10F2 that would be produced and label with a star all the chiral centers present in their structures.
The heat of combustion of decahydronaphthalene(C10H18) is -6286 kJ/mol. The heat of combustion ofnaphthalene (C10H8) is -5157 kJ/mol. (In both casesCO2(g) and H2O(l) are the products.) Calculate the heat of hydrogenationand the resonance energy of naphthalene.
Chapter 5 Solutions
BNDL: ACP ORGANIC CHEMISTRY:CH EM 231(W/ACCESS CARD)
Ch. 5.1 - Calculate the index of hydrogen deficiency for...Ch. 5.1 - Prob. 5.2PCh. 5.2 - Write the IUPAC name of each alkene. (a) (b)Ch. 5.2 - Prob. 5.4PCh. 5.2 - Prob. 5.5PCh. 5.2 - Prob. 5.6PCh. 5.2 - Prob. 5.7PCh. 5.2 - Prob. 5.8PCh. 5 - Predict all approximate bond angles about each...Ch. 5 - Prob. 5.10P
Ch. 5 - The structure of 1,2-propadiene (allene) is shown...Ch. 5 - Prob. 5.12PCh. 5 - Draw structural formulas for these alkenes. (a)...Ch. 5 - Name these alkenes and cycloalkenes.Ch. 5 - Prob. 5.15PCh. 5 - Prob. 5.16PCh. 5 - Prob. 5.17PCh. 5 - For each molecule that shows cis, trans isomerism,...Ch. 5 - -Ocimene, a triene found in the fragrance of...Ch. 5 - Prob. 5.20PCh. 5 - Prob. 5.21PCh. 5 - Prob. 5.22PCh. 5 - Prob. 5.23PCh. 5 - Prob. 5.24PCh. 5 - Measure the CH3,CH3 distance in the...Ch. 5 - Prob. 5.26PCh. 5 - Measure the CCC and CCH bond angles in the...Ch. 5 - Prob. 5.28PCh. 5 - Prob. 5.29PCh. 5 - Prob. 5.30PCh. 5 - Prob. 5.31PCh. 5 - Prob. 5.32PCh. 5 - Prob. 5.33PCh. 5 - Pyrethrin II and pyrethrosin are two natural...Ch. 5 - Prob. 5.35PCh. 5 - Prob. 5.36PCh. 5 - Bromine adds to cis- and trans-2-butene to give...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw structural formulas for the cis and trans isomers of hydrindane. Show each ring in its most stable conformation. Which of these isomers is more stable?arrow_forwardNew hydrocarbon compounds K, L, M and N have been formed from an organic reactions in Dr. Meredith laboratory. She found that compound K and L with molecular formula (C;H14) are unsaturated hydrocarbons with positional isomerism. Meanwhile, the reaction of hydrocarbon from compound M produces carbon dioxide and water when burns in plentiful oxygen. Another observation showed compound N produces three products X, Y and Z when react with chlorine gas in the presence of UV light. Identify the name of unsaturated hydrocarbon compound K and L and draw their possible condensed and skeletal structures. Suggest the IUPAC nomenclature name for compound M and write the complete chemical reaction of compound M when burns in plentiful oxygen. Show the complete mechanism reaction that occur for the formation of products X, Y and Z from compound N.arrow_forwardCalculate the units of unsaturation of C6H7NO. And draw out the structure.arrow_forward
- For each of the following compounds,determine whether each is optically active. For optically active compounds, identify the chiral carbon: ethane, 2-chloro-2-methylbutane, CH3CH(NH2)COOH, CH3CH2CHClCH3and CH3CH2CH2CH3. And for non-optical active compounds, just provide the structure of the compoundarrow_forwardDraw and name a constitutional isomer of C9H18 that has cis/trans isomers and is named a “3-hexene”. (Label your structure with the proper E/Z nomenclature).arrow_forwarddraw and name two structures that match the description of a trans-dihalocyclopentanearrow_forward
- Draw the structure(s) of all of the alkene isomers, C5H10, that contain a branched chain. Consider E/Z stereochemistry of alkenes.arrow_forward"A research team synthesizes a novel organic compound 'X' with the molecular formula C5H8O2. When 'X' is treated with a deuterated acid (D2O), a single deuterium atom replaces a hydrogen atom, forming compound 'Y' (C5H7DO2). 'X' does not react with 2,4-Dinitrophenylhydrazine (2,4-DNP) but does react with both Tollens' reagent and Benedict's solution, forming a silver mirror and a red precipitate, respectively. Furthermore, 'X' undergoes catalytic hydrogenation over a palladium catalyst, consuming one mole of hydrogen to form a compound 'Z' (C5H10O2). Based on these observations, what is the most likely structure of compound 'X'?" A. Methyl vinyl ketone B. 3-Buten-2-one C. Acetoacetic ester D. 2-Hydroxypent-3-enalarrow_forwardIt is easy to imagine a cyclohexane as a flat hexagon and a lot of the time we draw it that way. Looking at 1,3,5-triethylcyclohexane we cannot tell the stability of the molecule from looking at the flat 2D drawing. Explain why we need to look at the 3D configuration and what conformation (axial,equatorial) would each of the three ethyl groups be in for the most stable configuration.arrow_forward
- Compound X, C,4H12Br2, is optically inactive. On treatment with strong base, X gives hydrocarbon Y, C14H10: Compound Y absorbs 2 equivalents of hydrogen when reduced over a palladium catalyst to give z (C14H14) and reacts with ozone to give one product, benzoic acid (C,Hg02). Draw the structure of compound Z. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Ignore alkene stereochemistry. • If more than one structure fits the description, draw them all. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate structures with + signs from the drop-down menu. ChemDoodlearrow_forwardCompound W, C6H13Cl, undergoes base-promoted E2 elimination to give a single C6H₁2 alkene, Y. Compound X, C6H13Br, undergoes a similar reaction to form Y and an isomeric alkene Z. W is chiral; X is not. Catalytic hydrogenation of Y and Z produces 2-methylpentane. Propose structures for W and X. 0 • Do not use stereobonds in your answer. • In cases where there is more than one possible structure for each molecule, just give one for each. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu. -8) ChemDoodleⓇ Sn [Farrow_forwardAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent ofH2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragmentis propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are thestructures of A, B, and C? Write all reactions and show your reasoning.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
