
Modified Mastering Chemistry with Pearson eText -- Standalone Access Card -- for General, Organic, and Biological Chemistry (3rd Edition)
3rd Edition
ISBN: 9780134143705
Author: Laura D. Frost, S. Todd Deal
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.60AP
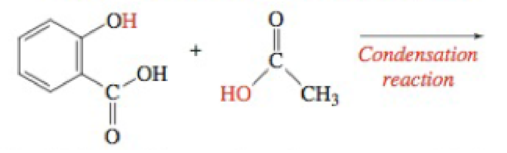
Acetylsalicylic acid (aspirin) can be synthesized by combining salicylic add and acetic acid through a condensation reaction. The —OH group from phenol on the salicylic acid condenses with the carboxylic acid group of acetic acid forming acctyl-salicylic add. Draw the structure of acetylsalicylic acid.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
What is the balanced equation for salicylic acid + acetic anhydride --> acetylsalicylic acid (aspirin) + acetic acid? When added, why is concentrated sulfuric acid a catalyst?
Write the products formed as a result of the given reactions.
Ethanol can be used as a fuel source in an alcohol lamp. The formula for ethanol is C2H5OH. Write the balanced equation for the combustion of ethanol.
Chapter 5 Solutions
Modified Mastering Chemistry with Pearson eText -- Standalone Access Card -- for General, Organic, and Biological Chemistry (3rd Edition)
Ch. 5 - When vinegar (CH3COOH) and baking soda (NaHCO3)...Ch. 5 - In your own words, define free energy change, G....Ch. 5 - Classify the following as exothermic or...Ch. 5 - Prob. 5.4PPCh. 5 - Prob. 5.5PPCh. 5 - Classify the following as spontaneous or...Ch. 5 - Prob. 5.7PPCh. 5 - Prob. 5.8PPCh. 5 - Prob. 5.9PPCh. 5 - Prob. 5.10PP
Ch. 5 - a. How does increasing the temperature increase...Ch. 5 - a. Describe activation energy for a chemical...Ch. 5 - Why does the rate of a chemical reaction decrease...Ch. 5 - Prob. 5.14PPCh. 5 - Enzymes increase the rate of a biological chemical...Ch. 5 - Prob. 5.16PPCh. 5 - Prob. 5.17PPCh. 5 - Prob. 5.18PPCh. 5 - Categorize the following reactions as synthesis,...Ch. 5 - Categorize the following reactions as synthesis,...Ch. 5 - Prob. 5.21PPCh. 5 - Prob. 5.22PPCh. 5 - Write the products and balance the following...Ch. 5 - Prob. 5.24PPCh. 5 - Prob. 5.25PPCh. 5 - List the differences between general chemical...Ch. 5 - Are the substances shown in italics undergoing...Ch. 5 - Prob. 5.28PPCh. 5 - Prob. 5.29PPCh. 5 - Prob. 5.30PPCh. 5 - Prob. 5.31PPCh. 5 - Prob. 5.32PPCh. 5 - Prob. 5.33PPCh. 5 - Prob. 5.34PPCh. 5 - Prob. 5.35PPCh. 5 - Prob. 5.36PPCh. 5 - Prob. 5.37PPCh. 5 - Write the main product of hydration for the...Ch. 5 - Methane (a.k.a. natural gas) can react with oxygen...Ch. 5 - Prob. 5.40APCh. 5 - Which reaction occurs at a faster rate, an...Ch. 5 - Prob. 5.42APCh. 5 - Prob. 5.43APCh. 5 - Two curves for the same reaction are shown in the...Ch. 5 - Prob. 5.45APCh. 5 - Draw and label a reaction energy diagram for an...Ch. 5 - Prob. 5.47APCh. 5 - Prob. 5.48APCh. 5 - Write the products that would result from the...Ch. 5 - Prob. 5.50APCh. 5 - Prob. 5.51APCh. 5 - Prob. 5.52APCh. 5 - Prob. 5.53APCh. 5 - Prob. 5.54APCh. 5 - Prob. 5.55APCh. 5 - Identify the reactant that is oxidized and the...Ch. 5 - Prob. 5.57APCh. 5 - Write the products of the following reactions:Ch. 5 - Prob. 5.59APCh. 5 - Acetylsalicylic acid (aspirin) can be synthesized...Ch. 5 - Prob. 5.61APCh. 5 - Fill in the missing organic produce for the...Ch. 5 - Prob. 5.63APCh. 5 - Prob. 5.64APCh. 5 - How do low-carb diets work? We store glucose...Ch. 5 - Prob. 5.66CPCh. 5 - Prob. 5.67CPCh. 5 - Prob. 5.68CPCh. 5 - Prob. 5.69CPCh. 5 - Which reaction has the larger activation energy?Ch. 5 - Prob. 1IA.2QCh. 5 - A catalyst speeds up a chemical reaction by...Ch. 5 - Prob. 1IA.4QCh. 5 - Examine your sketch from question 3. Does a...Ch. 5 - Prob. 1IA.6QCh. 5 - Prob. 2IA.1QCh. 5 - Prob. 2IA.2QCh. 5 - Prob. 2IA.3QCh. 5 - One of the reactions in the data set is a single...Ch. 5 - Prob. 2IA.5QCh. 5 - Categorize the following reactions as a synthesis,...Ch. 5 - Prob. 3IA.1QCh. 5 - Prob. 3IA.2QCh. 5 - Prob. 3IA.3QCh. 5 - Prob. 3IA.4QCh. 5 - Prob. 3IA.5QCh. 5 - Prob. 1ICCh. 5 - Find out how unsaturated fats are saturated and...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete and balance the reaction for the complete combustion of the following fuels. You do not need to indicate the physical state of the reactants and products Propane, C3H8 Butyl Alcohol, C4H9OHarrow_forwardAspirin can be made in the laboratory by reacting acetic anhydride (C4H6O3C4H6O3) with salicylic acid (C7H6O3C7H6O3) to form aspirin (C9H8O4C9H8O4) and acetic acid (C2H4O2C2H4O2). The balanced equation isC4H6O3+C7H6O3→C9H8O4+C2H4O2C4H6O3+C7H6O3→C9H8O4+C2H4O2In a laboratory synthesis, a student begins with 2.90 mLmL of acetic anhydride (density=1.08gml−1density=1.08gml−1) and 1.28 gg of salicylic acid. Once the reaction is complete, the student collects 1.20 gg of aspirin. a) Determine the theoretical yield of aspirin for the reaction. Express your answer using three significant figures. b) Determine the percent yield of aspirin for the reaction. Express your answer using three significant figures.arrow_forwardAspirin can be made in the laboratory by reacting acetic anhydride (C4H6O3C4H6O3) with salicylic acid (C7H6O3C7H6O3) to form aspirin (C9H8O4C9H8O4) and acetic acid (C2H4O2C2H4O2). The balanced equation isC4H6O3+C7H6O3→C9H8O4+C2H4O2C4H6O3+C7H6O3→C9H8O4+C2H4O2In a laboratory synthesis, a student begins with 3.10 mLmL of acetic anhydride (density=1.08gml−1density=1.08gml−1) and 1.23 gg of salicylic acid. Once the reaction is complete, the student collects 1.22 gg of aspirin. Determine the percent yield of aspirin for the reaction. Express your answer using three significant figures.arrow_forward
- The heat of combustion of glucose, C6H12O6, is 670 kcal/mol. The heat of combustion of ethanol, C2H6O, is 327 kcal/mol. The heat liberated by oxidation of each compound is the same whether is burned in the air or metabolized in the body. On a kcal/g basis, metabolism of which compound liberates more heat?arrow_forwardWhat is the reaction product?arrow_forwardFor some questions you will need to use the special periodic table attached in the images below! Treat Je, Qu, Ap, and Bg as NONMETALS! A) Enter the balanced chemical equation for the reaction of 2-bogusopropanoic acid with KOH. B) Enter the balanced equation for the ionization of 2-bogusopropanoic acid in water.arrow_forward
- Write the chemical equation for the reaction of ethanol with sodium metalarrow_forwardWhat type of reaction is Na +H2O —>arrow_forwardIn a combustion reaction, the most common fuel that burns contains compounds called hydrocarbons. This organic compound reacts quickly with oxygen and produces carbon dioxide (CO2) and water vapor (H2O). The reaction typically gives off heat and light as well. The combustion of methane is represented by the equation: CH4 + 2O2 → CO2 + 2H2O 1. In the above reaction what compound is oxidized? 2. Give another example of a hydrocarbon combustion reaction and write the equation.arrow_forward
- The organic lab program generates around 200 liters of non-halogenated organic waste every week. A student accidentally added some halogenated organic waste to a waste container that entered the non-halogenated waste stream. The cost to dispose of 1.000 liter of non-halogenated organic waste is $20.27 The cost to dispose of 1.000 liter of halogenated organic waste is $89.71 Due to the error, how much extra money will it cost to dispose of 210.9 liters of the waste that week?arrow_forwardWrite the chemical formulas for the products formed when each of the following hydrocarbons undergoes complete combustion. a. CH4 b. C4H10 c. C4H6 d. C6H6arrow_forwardWrite a chemical equation that involves water as a reactant for the formation of the a. water free radical b. hydroxyl free radicalarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY