
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
3rd Edition
ISBN: 9781259298424
Author: SMITH
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.77P
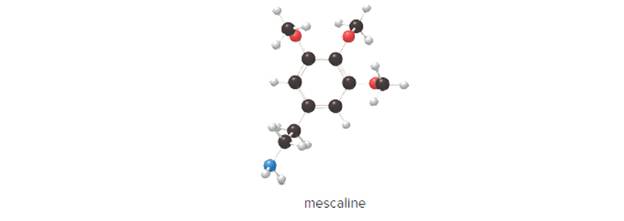
Mescaline is a hallucinogen in peyote, a cactus native to the southwestern UnitedStates and Mexico. (a) What is the chemical formula of mescaline? (b) Calculate itsmolar mass. (c) How many moles are contained in 7.50 g of mescaline?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) What is the chemical formula of eugenol, the main contri butor to the aroma of cloves? (b) Calculate its molar mass. (c) How many moles are contained in 10.50 g of eugenol?
Mescaline is a hallucinogen in peyote, a cactus native to the southwestern United States and Mexico. (a) What is the chemical formula of mescaline? (b) Calculate its molar mass. (c) How many moles are contained in 7.50 g of mescaline?
The molecular formula of allicin, the compound responsible for the characteristic smell of garlic, is C6H10OS2. (a) What is the molar mass of allicin? (b) How many moles of allicin are present in 5.00 mg of this substance?
Chapter 5 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Label the reactants and products, and indicate how...Ch. 5.1 - One term in a balanced chemical equation contained...Ch. 5.2 - Write a balanced equation for each reaction....Ch. 5.2 - Write a balanced equation for the following...Ch. 5.2 - Write a balanced equation for the reaction of...Ch. 5.2 - Balance each chemical equation. Al+H2SO4Al2(...Ch. 5.3 - Write a balanced equation for the reaction...Ch. 5.3 - Classify each reaction as a combination or...
Ch. 5.3 - Classify each reaction as a single replacement or...Ch. 5.3 - Classify each reaction as a combination,...Ch. 5.3 - Fill in the needed reactants or products for each...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - Prob. 5.16PCh. 5.5 - How many items are contained in one mole of (a)...Ch. 5.5 - How many carbon atoms are contained in each of the...Ch. 5.5 - How many molecules are contained in each of the...Ch. 5.5 - How many moles of water contain each of the...Ch. 5.6 - Calculate the formula weight of each ionic...Ch. 5.6 - The unmistakable odor of a freshly cut cucumber is...Ch. 5.6 - Prob. 5.23PCh. 5.6 - Calculate the number of grams contained in each of...Ch. 5.6 - How many moles are contained in each of the...Ch. 5.6 - How many molecules are contained in two 500.-mg...Ch. 5.7 - Use the balanced equation for the reaction of N2...Ch. 5.7 - Use the balanced equation in Sample Problem 5.15...Ch. 5.8 - Prob. 5.29PCh. 5.8 - Using the balanced equation for the combustion of...Ch. 5.8 - Prob. 5.31PCh. 5.8 - Use the balanced equation, N2+O22NO, to answer the...Ch. 5.9 - Prob. 5.33PCh. 5.9 - Prob. 5.34PCh. 5.9 - The synthetic antiviral drug Tamiflu, currently...Ch. 5.10 - Consider the reaction of hydrogen and nitrogen to...Ch. 5.10 - Using the balanced equation for the reaction of H2...Ch. 5.10 - Using the balanced equation, 3H2(g)+N2(g)2NH3(g),...Ch. 5.10 - Using the balanced equation, N2(g)+O2(g)2NO(g),...Ch. 5.10 - Prob. 5.41PCh. 5.10 - Prob. 5.42PCh. 5 - Prob. 5.43PCh. 5 - Prob. 5.44PCh. 5 - How many atoms of each element are drawn on each...Ch. 5 - How many atoms of each element are drawn on each...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Prob. 5.48PCh. 5 - Balance each equation. a....Ch. 5 - Balance each equation. a....Ch. 5 - Prob. 5.51PCh. 5 - Prob. 5.52PCh. 5 - Prob. 5.53PCh. 5 - Prob. 5.54PCh. 5 - For the reaction depicted in the molecular art:...Ch. 5 - Prob. 5.56PCh. 5 - Prob. 5.57PCh. 5 - Prob. 5.58PCh. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Prob. 5.67PCh. 5 - Rechargeablenickel-cadmium batteries are used in...Ch. 5 - The reaction of hydrogen (H2) with acetylene...Ch. 5 - Prob. 5.70PCh. 5 - Calculate the formula weight and molar mass of...Ch. 5 - Calculate the formula weight and molar mass of...Ch. 5 - L-Dopa is a drug used to treat Parkinson’s...Ch. 5 - Niacin, vitamin B3, is found in soybeans, which...Ch. 5 - Which quantity has the greater mass? 1 mol of Fe...Ch. 5 - Prob. 5.76PCh. 5 - Mescaline is a hallucinogen in peyote, a cactus...Ch. 5 - Prob. 5.78PCh. 5 - How many grams are contained in 5.00 mol of each...Ch. 5 - How many grams are contained in 0.50 mol of each...Ch. 5 - Prob. 5.81PCh. 5 - How many moles are contained in each number of...Ch. 5 - Prob. 5.83PCh. 5 - Prob. 5.84PCh. 5 - Prob. 5.85PCh. 5 - Prob. 5.86PCh. 5 - Using the balanced equation for the combustion of...Ch. 5 - Sodium metal (Na) reacts violently when added to...Ch. 5 - Prob. 5.89PCh. 5 - Prob. 5.90PCh. 5 - What is the percent yield of B in a reaction that...Ch. 5 - What is the percent yield of B in a reaction that...Ch. 5 - The reaction of methane (CH4) with Cl2forms...Ch. 5 - Methanol (CH4O), which is used as a fuel in...Ch. 5 - Consider the given reaction mixture that contains...Ch. 5 - Consider the reaction of A2 and B2 to form A2B,...Ch. 5 - Prob. 5.97PCh. 5 - Prob. 5.98PCh. 5 - Prob. 5.99PCh. 5 - Prob. 5.100PCh. 5 - The local anesthetic ethyl chloride ( C2H5Cl,...Ch. 5 - The solvent dichloromethane (, molar mass 84.93...Ch. 5 - Answer the following questions about the...Ch. 5 - Answer the following questions about diethyl ether...Ch. 5 - Prob. 5.105PCh. 5 - Prob. 5.106PCh. 5 - Prob. 5.107PCh. 5 - Prob. 5.108PCh. 5 - Prob. 5.109PCh. 5 - Prob. 5.110PCh. 5 - DDT, a pesticide that kills disease-carrying...Ch. 5 - Prob. 5.112PCh. 5 - TCDD, also called dioxin...Ch. 5 - Prob. 5.114CP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Which of the following solutions has the higher molarity? 10 ppm KI in water or 10,000 ppb KBr in water 0.25 ma...
CHEMISTRY-TEXT
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
What is the pH range for acidic solutions? For basic solutions?
Introduction to Chemistry
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 4.61 What is actually measured by the octane ratings of different grades of gasoline?arrow_forwardNitrogen fixation in the root nodules of peas and other legumes occurs with a reaction involving a molybdenum-containing enzyme named nitrogenase. This enzyme contains two Mo atoms per molecule and is 0.0872% Mo by mass. Calculate the molar mass of the enzyme.arrow_forward(a) Calculate the molar mass of ammonium nitrate, NH4NO3. (b) What is the mass percent of nitrogen in ammonium nitrate? (c) How many kilograms of nitrogen are present in a 10.0-lb bag of ammonium nitrate? (d) How many kilograms of ammonium nitrate contain 1.00 lb of nitrogen? (1 lb = 453.6 g)arrow_forward
- (a) What is the mass, in grams, of 4.62 1023 molecules of aspirin, C9H8O4? (b) What is the molar mass of diazepam (Valium(R)) if 0.05570 mol weighs 15.86 g? g/mol (c) How many moles of ammonium ions are in 0.645 g of ammonium carbonate?arrow_forwardA 1.30 g sample of titanium chemically combines with chlorine gas to form 5.16 g of titanium chloride. (a) What is the empirical formula of titanium chloride? (b) What is the percent by mass of titanium and the percent by mass of chloride in the sample?arrow_forwardA mixture of O2 (g) and H2 (g) explosively react to form steam H2O (g) in the presence of an electric spark. 4.15 mol of H2 (g) and 7.13 mol of O2 (g) are mixed and allowed to react in the presence of a electric spark. The yield of the reaction is 100%. (a) Write a balanced equation for the reaction. (b) What is the limiting reactant? (answer: 4.15 mol H2 can produce 4.15 mol H2O and 7.13 mol O2 can produce 14.3 mol of H2O, so, H2 is the limiting reactant.) (c) What is the theoretical yield of H2O in moles? (answer: 4.15 mol) (d) How many moles of O2 are left after the reaction? (answer: 5.05 mol) (e) How many moles of H2 and O2 would be left if the reaction yield is only 74.5%? (answer: H2: 1.06 mol left; O2: 5.59 mol)arrow_forward
- Consider the mixture of propane, C3H8, and O2 shown here.(a) Write a balanced equation for the combustion reaction thatoccurs between propane and oxygen. (b) Which reactant is thelimiting reactant? (c) How many molecules of CO2, H2O, C3H8,and O2 will be present if the reaction goes to completion?arrow_forwardCitric acid (C6H8O7) is made by fermentation of sugars such as sucrose (C12H22O11) in air. Oxygen is consumed and water generated as a by-product.(a) Write a balanced equation for the overall reaction that occurs in the manufacture of citric acid from sucrose.(b) What mass of citric acid is made from 15.0 kg sucrose?arrow_forward1.Ibuprofen is a compound used in painkillers. When a 2.174 g sample is burned in an excess of oxygen, it yields 6.029 g CO2 and 1.709 g H2O as the sole products. (a)What is the percent composition, by mass, of ibuprofen? (b) What is the empirical formula of ibuprofen?arrow_forward
- (a) What is the mass, in grams, of one mole of 12C? (b) Howmany carbon atoms are present in one mole of 12C?arrow_forward3) An organic compound contains only C, H, and O. Complete combustion of a 3.185 g sample in an excess of oxygen yields 8.846 g CO2 and 2.507 g H2O. (a) What is the percent composition, by mass, of this compound? (b) What is the empirical formula of the compound? (c) If the molar mass of this compound is 206 g/mol, what is the molecular formula? (C: 12.01 g/mol; H: 1.01 g/mol; O: 16.0 g/mol)arrow_forwardDDT, an insecticide harmful to fish, birds, and humans, is produced by the following reaction. 2 C6H5Cl + C2HOCl3 → C14H9Cl5 + H2O chlorobenzene chloral DDT In a government lab, 1169 g of chlorobenzene is reacted with 528 g of chloral. (a) What mass of DDT is formed, assuming 100% yield? g(b) Which reactant is limiting? C6H5Cl C2HOCl3 neither reactant is limiting Which is in excess? C6H5Cl C2HOCl3 neither reactant is in excess (c) What mass of the excess reactant is left over? g(d) If the actual yield of DDT is 188.2 g, what is the percent yield? %arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY