
ORGANIC CHEMISTRY-W/ACCESS
5th Edition
ISBN: 9781259993398
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.7P
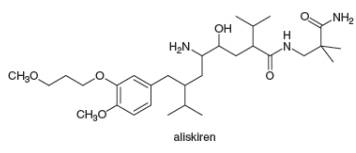
The facts in section 5.4A can be used to locate stereogenic centers in any molecule, no matter how complicated. Always look for carbons surrounded by four different groups. With this in mind, locate the four stereogenic centers in aliskiren, a drug introduced in
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The principles in Section 5.4A can be used to locate stereogenic centers in any molecule, no matter how complicated. Always look for carbons surrounded by four different groups. With this in mind, locate the four stereogenic centers in aliskiren, a drug introduced in 2007 for the treatment of hypertension.
Label them based on the type of stereoisomerism they exhibit in.
OPTICAL. NEITHER OPTICAL OR GEOMETRIC GEOMETRIC
CH3CH(CBr3)CH2CH2CH3
CHCI=CHCH2CI
CH3CH(CBr3)CH2CH3
CF2=CI2
CHBr=CHCH2Br
There are nine constitutional isomers of molecular formula C7H16.
1) What is the unsaturation number of each of these compounds?
2) Draw five constitutional isomers, where two of these are chiral compounds.
3) For the three achiral constitutional isomers, provide the correct IUPAC names.
4) For the two chiral constitutional isomers, draw in Fischer projection formula the enantiomers of each.
5) Name, according to IUPAC standards, each of the enantiomers you drew in #4 above.
Chapter 5 Solutions
ORGANIC CHEMISTRY-W/ACCESS
Ch. 5 - Prob. 5.1PCh. 5 - Prob. 5.2PCh. 5 - Draw the mirror image of each compound. Label each...Ch. 5 - Prob. 5.4PCh. 5 - A molecule is achiral if it has a plane of...Ch. 5 - Problem 5.6 Locate the stereogenic centers in each...Ch. 5 - The facts in section 5.4A can be used to locate...Ch. 5 - Prob. 5.8PCh. 5 - Prob. 5.9PCh. 5 - Prob. 5.10P
Ch. 5 - Prob. 5.11PCh. 5 - Prob. 5.12PCh. 5 - Label each compound as R or S.Ch. 5 - Prob. 5.14PCh. 5 - Prob. 5.15PCh. 5 - Prob. 5.16PCh. 5 - Prob. 5.17PCh. 5 - Problem 5.18 Compounds E and F are two isomers of...Ch. 5 - Prob. 5.19PCh. 5 - Prob. 5.20PCh. 5 - Prob. 5.21PCh. 5 - Prob. 5.22PCh. 5 - Prob. 5.23PCh. 5 - Prob. 5.24PCh. 5 - Which of the following cyclic molecules are meso...Ch. 5 - Prob. 5.26PCh. 5 - Prob. 5.27PCh. 5 - Problem 5.28 The amino acid has the physical...Ch. 5 - Prob. 5.29PCh. 5 - Prob. 5.30PCh. 5 - Prob. 5.31PCh. 5 - Prob. 5.32PCh. 5 - Prob. 5.33PCh. 5 - Prob. 5.34PCh. 5 - Prob. 5.35PCh. 5 - Prob. 5.36PCh. 5 - Prob. 5.37PCh. 5 - Prob. 5.38PCh. 5 - Prob. 5.39PCh. 5 - 5.40 Determine if each compound is identical to or...Ch. 5 - Prob. 5.41PCh. 5 - Prob. 5.42PCh. 5 - Prob. 5.43PCh. 5 - Prob. 5.44PCh. 5 - Prob. 5.45PCh. 5 - Prob. 5.46PCh. 5 - Label each stereogenic center as R or S. a. c. e....Ch. 5 - Prob. 5.48PCh. 5 - Prob. 5.49PCh. 5 - Prob. 5.50PCh. 5 - Prob. 5.51PCh. 5 - Prob. 5.52PCh. 5 - Prob. 5.53PCh. 5 - Prob. 5.54PCh. 5 - Prob. 5.55PCh. 5 - Prob. 5.56PCh. 5 - Prob. 5.57PCh. 5 - Prob. 5.58PCh. 5 - 5.59 Explain each statement by referring to...Ch. 5 - Prob. 5.60PCh. 5 - Prob. 5.61PCh. 5 - Prob. 5.62PCh. 5 - Prob. 5.63PCh. 5 - Prob. 5.64PCh. 5 - Prob. 5.65PCh. 5 - Prob. 5.66PCh. 5 -
5.67 Artemisinin and mefloquine are widely used...Ch. 5 - 5.68 Saquinavir (trade name Invirase) is a...Ch. 5 - Prob. 5.69PCh. 5 - Prob. 5.70PCh. 5 - Prob. 5.71PCh. 5 - Prob. 5.72PCh. 5 - Problem 5.73 An acid-base reaction of with a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw a three-dimensional structure for each compound. Denote the asymmetric carbon atom with an asterisk (*). Draw the mirror image for each structure, and state whether you have drawn a pair of enantiomers of just the same molecule twice.arrow_forwardShown below is Streptomycin, an antibiotic medication used to treat a number of bacterial infections, including tuberculosis, plague, and endocarditis. Neomycin B has broad-spectrum antibacterial activity. Circle and label as many functional groups in these molecules as you can. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?arrow_forwardGlucose is a simple sugar with five substituents bonded to a sixmembered ring.a.Using a chair representation, draw the most stable arrangement of these substituents on the six-membered ring. b.Convert this representation to one that uses a hexagon with wedges and dashed wedges. c.Draw a constitutional isomer of glucose. d.Draw a stereoisomer that has an axial OH group on one carbon.arrow_forward
- Glucose is a simple sugar with five substituents bonded to a six-membered ring.a. Using a chair representation, draw the most stable arrangement of these substituents on the six-membered ring.b. Convert this representation into one that uses a hexagon with wedges and dashed wedges.c. Draw a constitutional isomer of glucose.d. Draw a stereoisomer that has an axial OH group on one carbon.arrow_forwardShown below is Streptomycin, and Neomycin B. Circle and label as many functional groups in these molecules as you can. a. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? b. Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?arrow_forwardList down 5 enantiomer chemicals that are important in your daily life with their enantiomeric naming system, and what are both of their optical activity in R and S configuration with (angle of rotation). List them in a table.arrow_forward
- Label the following pairs as A) distereoisomers, B) enantiomers, C) conformers, or D) the same. b1 c1 d1 e1 a2 b2 c2 The structures above are labeled a1, a2, b1, b2 ...etc. Using these labels, indicate which compounds above would have an optical rotation ofarrow_forwardDefine Stereogenic Centers in Cyclic Compounds ?arrow_forwardNeuroprotectin D1 (NPD1) is synthesized in the body from highly unsaturated essential fatty acids. NPD1 is a potent natural anti-inflammatory agent. a.) Label each carbon–carbon double bond as conjugated or isolated.b.) Label each double bond as E or Z.c.) For each conjugated system, label the given conformation as s-cis or s-transarrow_forward
- Neuroprotectin D1 (NPD1) is synthesized in the body from highly unsaturated essential fatty acids. NPD1 is a potent natural anti-inflammatory agent.a.Label each carbon–carbon double bond as conjugated or isolated. b.Label each double bond as E or Z. c.For each conjugated system, label the given conformation as s-cis or s-trans.arrow_forwardConsider the compound below. a) Draw the structure showing stereochemistry, in which carbon 1 has S configuration and carbon 2 has R configuration. b) Draw the structure showing stereochemistry, in which carbons 1 and 2 have S configuration. c) are the two structures from part a and b diastereomers, identical, enantiomers, or unrelated?arrow_forwardUsing the cyclohexane with the C's numbered as shown, draw a chair form that fits each description. a.) The ring has an axial CH3 group at C1 and an equatorial OH on C2.b.) The ring has an equatorial CH3 group on C6 and an axial OH group on C4.c.) The ring has equatorial OH groups on C1, C2, and C5.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License