
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 5.3, Problem 6P
Interpretation Introduction
Interpretation:
The specific rotation of coniine is to be determined.
Concept introduction:
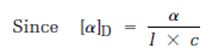
By following this mathematical formula, the specific rotation of the following molecule is to be determined.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
After creating her standard curve for absorption versus the concentration (M) of FD&C Red 40, a student found that her best fit linear line for FD&C Red 40 was y = 2,681x + -0.002.
Her Kool-Aid sample had an absorbance of 0.337. If 0.55 grams of Kool-Aid powder was used to prepare an 8-fl oz cup of her assigned flavor, what is the percent by mass of FD&C Red 40 in her 8-fl oz cup?
Calculate the wavenumber of the C-Cl band in CCl4.
what are the significant peaks of C4H9Br at
2970 cm-1
1209 cm-1
Chapter 5 Solutions
Organic Chemistry
Ch. 5.2 - Prob. 1PCh. 5.2 - Prob. 2PCh. 5.2 - Prob. 3PCh. 5.2 - Prob. 4PCh. 5.3 - Is cocaine (Worked Example 5-2) dextrorotatory or...Ch. 5.3 - Prob. 6PCh. 5.5 - Prob. 7PCh. 5.5 - Prob. 8PCh. 5.5 - Prob. 9PCh. 5.5 - Assign R or S configuration to the chirality...
Ch. 5.5 - Draw a tetrahedral representation of...Ch. 5.5 - Prob. 12PCh. 5.6 - One of the following molecules (a)–(d) is...Ch. 5.6 - Prob. 14PCh. 5.6 - Assign R or S configuration to each chirality...Ch. 5.7 - Prob. 16PCh. 5.7 - Which of the following have a meso form? (Recall...Ch. 5.7 - Does the following structure represent a meso...Ch. 5.8 - Prob. 19PCh. 5.8 - Prob. 20PCh. 5.9 - Prob. 21PCh. 5.11 - Prob. 22PCh. 5.11 - Prob. 23PCh. 5.11 - The lactic acid that builds up in tired muscles is...Ch. 5.11 - The aconitase-catalyzed addition of water to...Ch. 5.SE - Which of the following structures are identical?...Ch. 5.SE - Prob. 27VCCh. 5.SE - Prob. 28VCCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 30VCCh. 5.SE - Prob. 31APCh. 5.SE - Which of the following compounds are chiral? Draw...Ch. 5.SE - Prob. 33APCh. 5.SE - Eight alcohols have the formula C5H12O. Draw them....Ch. 5.SE - Draw compounds that fit the following...Ch. 5.SE - Prob. 36APCh. 5.SE - Prob. 37APCh. 5.SE - Prob. 38APCh. 5.SE - What is the stereochemical configuration of the...Ch. 5.SE - Prob. 40APCh. 5.SE - Prob. 41APCh. 5.SE - Prob. 42APCh. 5.SE - Prob. 43APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 46APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configurations to the chirality...Ch. 5.SE - Assign R or S stereochemistry to the chirality...Ch. 5.SE - Prob. 50APCh. 5.SE - Draw examples of the following: (a) A meso...Ch. 5.SE - Prob. 52APCh. 5.SE - Prob. 53APCh. 5.SE - Prob. 54APCh. 5.SE - On reaction with hydrogen gas by a platinum...Ch. 5.SE - Prob. 56APCh. 5.SE - Prob. 57APCh. 5.SE - One of the steps in fat metabolism is the...Ch. 5.SE - The dehydration of citrate to yield cis-aconitate,...Ch. 5.SE - The first step in the metabolism of glycerol,...Ch. 5.SE - One of the steps in fatty-acid biosynthesis is the...Ch. 5.SE - Prob. 62APCh. 5.SE - Draw tetrahedral representations of the two...Ch. 5.SE - The naturally occurring form of the amino acid...Ch. 5.SE - Prob. 65APCh. 5.SE - Prob. 66APCh. 5.SE - Prob. 67APCh. 5.SE - Allenes are compounds with adjacent carbon-carbon...Ch. 5.SE - Prob. 69APCh. 5.SE - Prob. 70APCh. 5.SE - How many stereoisomers of...Ch. 5.SE - Draw both cis- and trans-1,4-dimethylcyclohexane...Ch. 5.SE - Draw both cis- and trans-1,3-dimethylcyclohexane...Ch. 5.SE - cis-1,2-Dimethylcyclohexane is optically inactive...Ch. 5.SE - Prob. 75APCh. 5.SE - Prob. 76APCh. 5.SE - Prob. 77AP
Knowledge Booster
Similar questions
- Calculate the specific rotation of a solution of a compound (30.0 g/100 mL of solution) that has a measured rotation of +25.25º in a 2.00 dm tube.arrow_forwardA proton signal measured in a 90MHz spectrometer shows a doublet with peaks at 5.65 and 5.72 ppm. What is the chemical shift and coupling constant for this doublet? a. δ=5.69ppm, J=5.17 Hz b. δ=5.69ppm, J=6.30 Hz c. δ=5.72ppm, J=6.30 Hz d. δ=5.72ppm, J=5.17 Hzarrow_forwardAn ethanol solution of 3.5 mg/100 ml of compound Y (150.0 g/mol) in a 1.00 cm quartz cell has an extinction coefficient (ε) of 23,150 at λmax=235 nm. What is the expected absorption (A)? Report your answer to the correct number of significant figures.arrow_forward
- Propanone (acetone, (CH3)2CO) has a strong absorption at 189 nm and a weaker absorption at 280 nm. Identify the chromophore and assign the absorptions to π* ← n or π* ← π transitions.arrow_forwardThe natural abundance of 13C is only 1.1%. Furthermore, its sensitivity in NMR spectroscopy (a measure of the energy difference between a spin aligned with or against an applied magnetic field) is only 1.6% that of 1H. What are the relative signal intensities expected for the 1H-NMR and 13C-NMR spectra of the same sample of Si(CH3)4?arrow_forwardIf the measured absorbance at 280 nm of the protein in a 1.00-cm cuvette = 0.564, what is the extinction coefficient at this wavelength?arrow_forward
- Why is it most accurate to measure absorbances in the range A = 0.3 to 2?arrow_forwardThe Rf values for several dyes in an ink are listed below. Which dye has the greatest affinity for the stationary phase? Dye #1: Rf = 0.263 Dye #2: Rf = 0.551 Dye #3: Rf = 0.737 Dye #4: Rf = 0.882arrow_forwardPredict the relative intensities of the M and M+ 2 peaks for the following. Q) BrCH2CH2Brarrow_forward
- 50 uL of an aqueous sample of double-stranded DNA is dissolved in 950 uL of water. This diluted solution has a maximal absorbance of 0.326 at 260 nm. What is the concentration of the original (more concentrated) DNA sample, expressed in ug/uL?arrow_forwardHow do I find the [FeSCN]^2+ value?arrow_forwardWhat is the concentration of a tyrosine solution when the absorbance at 280 nm is 1.5 using a 1 cm cuvette and the molar extinction coefficient is 1405 L/mol·cm?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning