
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
4th Edition
ISBN: 9781260269284
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
thumb_up100%
Chapter 5.4, Problem 5.9P
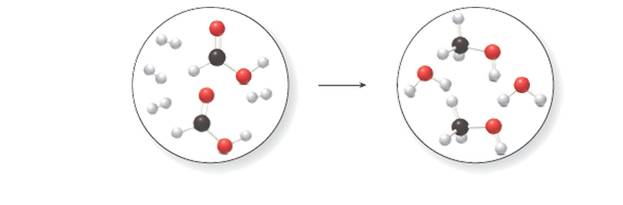
(a) Write a balanced equation for the reaction depicted in the molecular art.
(b) Identify the species that is oxidized and the species that is reduced.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A solid piece of aluminum is added to a test tube containing a solution of iron (II) chlorate [Fe(ClO3)2]. The piece of aluminum begins to darken in color, and the test tube becomes warm to the touch (evidence of chemical change).
(a) write the balanced molecular equation that happened in the test tube.
(b) which metal was oxidized
(c) which metal was reduced
The oxidation number of Cl in HClO₂ is:
For each chemical reaction listed in the table below, decide whether the highlighted atom is being oxidized or reduced.
highlighted atom is being...
reaction
neither
oxidized
reduced
oxidized nor
reduced
2 H2(9)+O2(9) → 2 H,0(9)
4 HF (9)+ SiO2(s) –
→ SiF,(9)+2 H20(g)
C(s)+O2(9) → CO2(9)
CH,(9)+H,O(9) →CO(g)+3 H,(9)
Chapter 5 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Use the molecular art to identify the process as a...Ch. 5.1 - Label the reactants and products, and indicate how...Ch. 5.1 - One term in a balanced chemical equation contained...Ch. 5.2 - Write a balanced equation for each reaction....Ch. 5.2 - Write a balanced equation for the following...Ch. 5.2 - Write a balanced equation for the reaction of...Ch. 5.2 - Consider the decomposition of hydrogen peroxide...Ch. 5.2 - Balance each chemical equation. Al+H2SO4Al2(...Ch. 5.3 - Write a balanced equation for the reaction...
Ch. 5.3 - Classify each reaction as a combination or...Ch. 5.3 - Classify each reaction as a single replacement or...Ch. 5.3 - Classify each reaction as a combination,...Ch. 5.3 - Fill in the needed reactants or products for each...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - Identify the species that is oxidized and the...Ch. 5.4 - (a) Write a balanced equation for the reaction...Ch. 5.4 - Prob. 5.10PCh. 5.5 - How many items are contained in one mole of (a)...Ch. 5.5 - How many carbon atoms are contained in each of the...Ch. 5.5 - How many molecules are contained in each of the...Ch. 5.5 - How many moles of water contain each of the...Ch. 5.6 - Calculate the formula weight of each ionic...Ch. 5.6 - The unmistakable odor of a freshly cut cucumber is...Ch. 5.6 - Prob. 5.11PPCh. 5.6 - Calculate the number of grams contained in each of...Ch. 5.6 - How many moles are contained in each of the...Ch. 5.6 - How many moles are contained in a 1,000.-mg dose...Ch. 5.6 - How many molecules are contained in two 500.-mg...Ch. 5.7 - Use the balanced equation for the reaction of N2...Ch. 5.7 - Use the balanced equation in Sample Problem 5.15...Ch. 5.8 - Prob. 5.16PPCh. 5.8 - Using the balanced equation for the combustion of...Ch. 5.8 - Prob. 5.17PPCh. 5.8 - Use the balanced equation, N2+O22NO, to answer the...Ch. 5.9 - What is the percent yield of X in a reaction that...Ch. 5.9 - Using the chemical equation in Sample Problem...Ch. 5.9 - Using the equation in Sample Problem 5.20, answer...Ch. 5.9 - The synthetic antiviral drug Tamiflu, currently...Ch. 5.10 - Consider the reaction of hydrogen and nitrogen to...Ch. 5.10 - Using the balanced equation for the reaction of H2...Ch. 5.10 - Using the balanced equation, 3H2(g)+N2(g)2NH3(g),...Ch. 5.10 - Using Sample Problem 5.21 as a guide, complete the...Ch. 5.10 - Using the balanced equation, N2(g)+O2(g)2NO(g),...Ch. 5.10 - Prob. 5.24PPCh. 5 - Prob. 23PCh. 5 - Prob. 24PCh. 5 - How many atoms of each element are drawn on each...Ch. 5 - How many atoms of each element are drawn on each...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Use the molecular art to write a balanced equation...Ch. 5 - Some coal is high in sulfur (S) content, and when...Ch. 5 - Balance each equation. a....Ch. 5 - Balance each equation. a....Ch. 5 - Prob. 33PCh. 5 - Prob. 34PCh. 5 - Prob. 35PCh. 5 - Prob. 36PCh. 5 - For the reaction depicted in the molecular art:...Ch. 5 - Answer the questions in Problem 5.37 for the...Ch. 5 - Prob. 39PCh. 5 - Prob. 40PCh. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify the reaction depicted in the molecular...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Classify each reaction as combination,...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Fill in the needed reactant or product for each of...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Identify the species that is oxidized and the...Ch. 5 - Prob. 49PCh. 5 - Rechargeablenickel-cadmium batteries are used in...Ch. 5 - The reaction of hydrogen (H2) with acetylene...Ch. 5 - Prob. 52PCh. 5 - Calculate the formula weight and molar mass of...Ch. 5 - Calculate the formula weight and molar mass of...Ch. 5 - L-Dopa is a drug used to treat Parkinson’s...Ch. 5 - Niacin, vitamin B3, is found in soybeans, which...Ch. 5 - Which quantity has the greater mass? 1 mol of Fe...Ch. 5 - Prob. 58PCh. 5 - Mescaline is a hallucinogen in peyote, a cactus...Ch. 5 - Prob. 60PCh. 5 - How many grams are contained in 5.00 mol of each...Ch. 5 - How many grams are contained in 0.50 mol of each...Ch. 5 - A bottle of the pain reliever ibuprofen (C13H18O2,...Ch. 5 - One dose of Maalox contains 500. mg each of...Ch. 5 - How many moles are contained in each number of...Ch. 5 - How many moles are contained in each number of...Ch. 5 - How many molecules of butane (C4H10) are contained...Ch. 5 - Prob. 68PCh. 5 - The average nicotine (C10H14N2, molar mass 162.3...Ch. 5 - How many moles of sucrose...Ch. 5 - What is the mass in grams of each quantity of...Ch. 5 - What is the mass in grams of each quantity of...Ch. 5 - Using the balanced equation for the combustion of...Ch. 5 - Sodium metal (Na) reacts violently when added to...Ch. 5 - Prob. 75PCh. 5 - Using the balanced equation for the reaction of Na...Ch. 5 - What is the percent yield of B in a reaction that...Ch. 5 - What is the percent yield of B in a reaction that...Ch. 5 - The reaction of methane (CH4) with Cl2forms...Ch. 5 - Methanol (CH4O), which is used as a fuel in...Ch. 5 - Consider the given reaction mixture that contains...Ch. 5 - Consider the reaction of A2 and B2 to form A2B,...Ch. 5 - Prob. 83PCh. 5 - Consider the reaction with the balanced equation,...Ch. 5 - Using the balanced equation, 2NO+O22NO2, determine...Ch. 5 - Prob. 86PCh. 5 - Prob. 87PCh. 5 - Completer the followin table using the given...Ch. 5 - The local anesthetic ethyl chloride...Ch. 5 - The solvent dischloromethane...Ch. 5 - Answer the following questions about the...Ch. 5 - Answer the following questions about diethyl ether...Ch. 5 - Prob. 93PCh. 5 - Prob. 94PCh. 5 - Prob. 95PCh. 5 - Prob. 96PCh. 5 - TCDD, also called dioxin...Ch. 5 - Prob. 98CP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, & Biological Chemistry
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
The chapter sections to review are shown in parentheses at the end of each problem. A "chemical-free” shampoo i...
Basic Chemistry
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Why is it that when something gains electrons, it is said to be reduced? What is being reduced?arrow_forward4.3 Explain the difference between complete and incomplete combustion.arrow_forwardWhen elemental copper is placed in a solution of silver nitrate, the following oxidationreduction reaction takes place. Forming elemental silver: :math>Cu(s)+2AgNO3(aq)Cu(NO3)2(aq)+2Ag(s) at mass of copper is required to remove all the silver from a silver nitrate solution containing 1.95 mg of silver nitrate?arrow_forward
- The respiration reaction is shown below. Identify the oxidizing agent. C6H12O66O26CO2+6H2O a.C6H12O6b.O2c.CO2d.H2Oarrow_forwardAnswer true or false. (a) When a substance is oxidized, it loses electrons. (b) When a substance gains electrons, it is reduced. (c) In a redox reaction, the oxidizing agent becomes reduced. (d) In a redox reaction, the reducing reagent be comes oxidized. (e) When Zn is converted to Zn2 ion, zinc is oxidized. (f) Oxidation can also be defined as the loss of oxygen atoms and/or the gain of hydrogen atoms. (g) Reduction can also be defmed as the gain of oxygen atoms and/or the loss of hydrogen atoms. (h) When oxygen, O2, is converted to hydrogen per oxide, H2O2, we say that O2 is reduced. (i) Hydrogen peroxide, H2O2, is an oxidizing agent. (j) All combustion reactions are redox reactions. (k) The products of complete combustion (oxidation) of hydrocarbon fuels are carbon dioxide, water, and heat. (l) In the combustion of hydrocarbon fuels, oxygen is the oxidizing agent and the hydrocarbon fuel is the reducing agent. (m) Incomplete combustion of hydrocarbon fuels can produce significant…arrow_forward8. Consider the following net ionic equation for a redox reaction: CHEM 1F92 Assignment 4. Stoichiometry, Oxidation-Reduction 2 NO3 + 3 Cu + 8 H → 2 NO + 3 Cu²+ + 4 H₂O (a) Which reactant gets oxidized? (c) Which reactant is the oxidizing agent? (b) Which reactant gets reduced? (d) Which reactant is the reducing agent?arrow_forward
- In Mg and HCl what is the balanced equation, ionic equation, net ionic equation and which element was oxidize and which element was reduced?arrow_forwardFor each chemical reaction listed in the table below, decide whether the highlighted atom is being oxidized or reduced. highlighted atom is being... reaction neither oxidized reduced oxidized nor reduced FeO(s)+CO(g) → Fe(s)+CO,(9) 2 NH3(aq)+H,SO4(aq) → (NH,),SO4(aq) C(s)+O,(9) → C0,(9) 4 KI(aq)+2 CuCl,(aq) → 2 CuI(s)+I,(aq)+4 KCl(aq)arrow_forwardIf you complete and balance the following oxidation-reduction reaction in basic solution NO2 -1aq2 + Al1s2 ¡ NH31aq2 + Al1OH24 -1aq2 how many hydroxide ions are there in the balanced equation (for the reaction balanced with the smallest whole-number coefficients)? (a) One on the reactant side (b) One on the product side (c) Four on the reactant side (d) Seven on the product side (e) Nonearrow_forward
- (a) Write the balanced molecular and net ionic equations forthe reaction between magnesium and cobalt(II) sulfate.(b) What is oxidized, and what is reduced in the reaction?arrow_forwardFor each chemical reaction listed in the table below, decide whether the highlighted atom is being oxidized or reduced. reaction highlighted atom is being... oxidized reduced neither oxidized nor reduced Na2CO3 (s) + H3PO4 (aq) → Na2HPO4 (aq) + CO2 (g) + H2O (l) 4HF (g) + SiO2 (s) → SiF4 (g) + 2H2O (g) NH3 (aq) + 2O2 (g) → HNO3 (aq) + H2O (l) 4KI (aq) + 2CuCl2 (aq) → 2CuI (s) + I2 (aq) + 4KCl (aq)arrow_forwardNonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Introduction to Electrochemistry; Author: Tyler DeWitt;https://www.youtube.com/watch?v=teTkvUtW4SA;License: Standard YouTube License, CC-BY