
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 5.SE, Problem 46AP
Interpretation Introduction
Interpretation:
The tetrahedral representation of the following molecules is to be done.
Concept introduction:
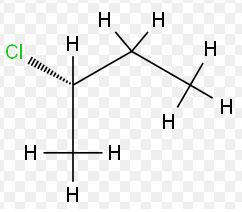
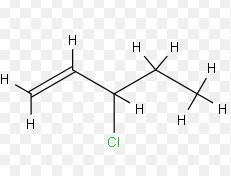
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw five isomers with the formula C8H18.
2. Draw the two chair conformations of menthol and identify the more stable conformation.
3. The pKa,1 and pKa,2 of ortho and para isomers of phthalic acid (diprotic acid) are shown below:
Provide an explanation on the differences of their pKa values.
The chiral catalyst (R)-BINAP-Ru is used to hydrogenate alkenes to give alkanes . The products are produced with high enantiomeric excess. An example is the formation of (S)-naproxen, a pain reliever.
Q.How can one enantiomer of naproxen be formed in such high yield?
1.By referring to the structure of 1-octen-3-ol, deet, citriodiol, icaridin and IR3535,a) Identify the functional groups and homologous series in each compound.
Chapter 5 Solutions
Organic Chemistry
Ch. 5.2 - Prob. 1PCh. 5.2 - Prob. 2PCh. 5.2 - Prob. 3PCh. 5.2 - Prob. 4PCh. 5.3 - Is cocaine (Worked Example 5-2) dextrorotatory or...Ch. 5.3 - Prob. 6PCh. 5.5 - Prob. 7PCh. 5.5 - Prob. 8PCh. 5.5 - Prob. 9PCh. 5.5 - Assign R or S configuration to the chirality...
Ch. 5.5 - Draw a tetrahedral representation of...Ch. 5.5 - Prob. 12PCh. 5.6 - One of the following molecules (a)–(d) is...Ch. 5.6 - Prob. 14PCh. 5.6 - Assign R or S configuration to each chirality...Ch. 5.7 - Prob. 16PCh. 5.7 - Which of the following have a meso form? (Recall...Ch. 5.7 - Does the following structure represent a meso...Ch. 5.8 - Prob. 19PCh. 5.8 - Prob. 20PCh. 5.9 - Prob. 21PCh. 5.11 - Prob. 22PCh. 5.11 - Prob. 23PCh. 5.11 - The lactic acid that builds up in tired muscles is...Ch. 5.11 - The aconitase-catalyzed addition of water to...Ch. 5.SE - Which of the following structures are identical?...Ch. 5.SE - Prob. 27VCCh. 5.SE - Prob. 28VCCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 30VCCh. 5.SE - Prob. 31APCh. 5.SE - Which of the following compounds are chiral? Draw...Ch. 5.SE - Prob. 33APCh. 5.SE - Eight alcohols have the formula C5H12O. Draw them....Ch. 5.SE - Draw compounds that fit the following...Ch. 5.SE - Prob. 36APCh. 5.SE - Prob. 37APCh. 5.SE - Prob. 38APCh. 5.SE - What is the stereochemical configuration of the...Ch. 5.SE - Prob. 40APCh. 5.SE - Prob. 41APCh. 5.SE - Prob. 42APCh. 5.SE - Prob. 43APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 46APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configurations to the chirality...Ch. 5.SE - Assign R or S stereochemistry to the chirality...Ch. 5.SE - Prob. 50APCh. 5.SE - Draw examples of the following: (a) A meso...Ch. 5.SE - Prob. 52APCh. 5.SE - Prob. 53APCh. 5.SE - Prob. 54APCh. 5.SE - On reaction with hydrogen gas by a platinum...Ch. 5.SE - Prob. 56APCh. 5.SE - Prob. 57APCh. 5.SE - One of the steps in fat metabolism is the...Ch. 5.SE - The dehydration of citrate to yield cis-aconitate,...Ch. 5.SE - The first step in the metabolism of glycerol,...Ch. 5.SE - One of the steps in fatty-acid biosynthesis is the...Ch. 5.SE - Prob. 62APCh. 5.SE - Draw tetrahedral representations of the two...Ch. 5.SE - The naturally occurring form of the amino acid...Ch. 5.SE - Prob. 65APCh. 5.SE - Prob. 66APCh. 5.SE - Prob. 67APCh. 5.SE - Allenes are compounds with adjacent carbon-carbon...Ch. 5.SE - Prob. 69APCh. 5.SE - Prob. 70APCh. 5.SE - How many stereoisomers of...Ch. 5.SE - Draw both cis- and trans-1,4-dimethylcyclohexane...Ch. 5.SE - Draw both cis- and trans-1,3-dimethylcyclohexane...Ch. 5.SE - cis-1,2-Dimethylcyclohexane is optically inactive...Ch. 5.SE - Prob. 75APCh. 5.SE - Prob. 76APCh. 5.SE - Prob. 77AP
Knowledge Booster
Similar questions
- One of the following molecules (a)–(d) is d-erythrose 4-phosphate, an intermediate in the Calvin photosynthetic cycle by which plants incorporate CO2 into carbohydrates. If d-erythrose 4-phosphate has R stereochemistry at both chirality centers, which of the structures is it? Which of the remaining three structures is the enantiomer of d-erythrose 4-phosphate, and which are diastereomers?arrow_forwardWhat is the stereochemical configuration of the enantiomer of (2S, 4R)-2, 4-octanediol? (A diol is a compound with two —OH groups.)arrow_forwardAllenes are compounds with adjacent carbon-carbon double bonds. Many allenes are chiral, even though they don’t contain chirality centers. Mycomycin, for example, a naturally occurring antibiotic isolated from the bacterium Nocardia acidophilus, is chiral and has [α]D = -130. Explain why mycomycin is chiral.arrow_forward
- Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forwardLithocholic acid is an A–B cis steroid found in human bile. Draw lithocholic acid showing chair conformations, as in Figure 27-11, and tell whether the hydroxyl group at C3 is axial or equatorial.arrow_forwardRank the following groups in order of decreasing priority. −F, −NH2, −CH3, −OHarrow_forward
- DHA is a fatty acid derived from sh oil and an abundant fatty acid in vertebrate brains. Hydrogenation of DHA forms docosanoic acid [CH3(CH2)20CO2H] and ozonolysis forms CH3CH2CHO, CH2(CHO)2 (ve equivalents), and HCOCH2CH2CO2H. What is the structure of DHA if all double bonds have the Z conguration?arrow_forward(a) Draw a three-dimensional line structure of (5R,6Z)-6,7-dichloro-5-hydroxynon-6-enal. Place the aldehyde group on the right-hand side of your drawing.(b) Give the Cahn-Ingold-Prelog priority order of the four groups (-OH, -H, -(CH2)3CHO, -C4H5Cl2) at the C5 stereocentre (Highest 1 > 2 > 3 > 4 Lowest).arrow_forwardProvide products for the following reactions. Label them as chiral, achiral, meso, dl, cis/trans, and stereocenters as R,S.arrow_forward
- Treatment of -D-glucose with methanol in the presence of an acid catalyst converts it into a mixture of two compounds called methyl glucosides (Section 25.3A). In these representations, the six-membered rings are drawn as planar hexagons. (a) Propose a mechanism for this conversion and account for the fact that only the OH on carbon 1 is transformed into an OCH3 group. (b) Draw the more stable chair conformation for each product. (c) Which of the two products has the chair conformation of greater stability? Explain.arrow_forwardWould 7b show stereochemistry? It looks like it has 2 chiral centers.arrow_forward(b) (i) Using Fischer projection, draw all the stereoisomers of 3-chloro-2-hydroxypentanoic acid. (ii) Assign the chiral centre(s) with R/S configurations in part (b)(i). Indicate which are enantiomers, diastereomers and meso compounds (if any).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning