
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
9th Edition
ISBN: 9781305780170
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.SE, Problem 55AP
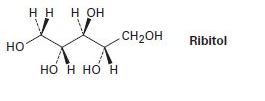
On reaction with hydrogen gas by a platinum catalyst, ribose (Problem 5-54) is converted into ribitol. Is ribitol optically active or inactive? Explain.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Identify the organic functional group(s) of the reactant, the reaction type, and predict the functional group(s) of the product(s) then draw the major product(s)
The reactant is:a. deoxytetroseb. alcohol pentosec. ketohexosed. aldotriosee. aldopentoseThe reaction type is:a. acetal formationb. oxidation (benedict's)c. hemiacetal formationd. acetal hydrolysise. reduction (hydrogenation)f. mutarotationThe product should be:a. alpha 1-4 disaccharideb. deoxyhexosec. carboxylic acid pentosed. alcohol pentosee. alpha pyranosef. no reaction
Citrate synthase, one of the enzymes in the series of enzyme-catalyzed reactions known as the citric acid cycle, catalyzes the synthesis of citric acid from oxaloacetic acid and acetyl-CoA. If the synthesis is carried out with acetyl-CoA that contains radioactive carbon (14C) in the indicated position , the isomer shown here is obtained. a. Which stereoisomer of citric acid is synthesized: R or S? b. If the acetyl-CoA used in the synthesis contains 12C instead of 14C, will the product of the reaction be chiral or achiral?
Trehalose, C12H22O11, is a nonreducing sugar that is only 45% as sweet as sugar. When hydrolyzed by aqueous acid or the enzyme maltase, it formsonly d-glucose. When it is treated with excess methyl iodide in the presence of Ag2O and then hydrolyzed with water under acidic conditions, only2,3,4,6-tetra-O-methyl-d-glucose is formed. Draw the structure of trehalose.
Chapter 5 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
Ch. 5.2 - Prob. 1PCh. 5.2 - Prob. 2PCh. 5.2 - Prob. 3PCh. 5.2 - Prob. 4PCh. 5.3 - Is cocaine (Worked Example 5-2) dextrorotatory or...Ch. 5.3 - Prob. 6PCh. 5.5 - Prob. 7PCh. 5.5 - Prob. 8PCh. 5.5 - Prob. 9PCh. 5.5 - Assign R or S configuration to the chirality...
Ch. 5.5 - Draw a tetrahedral representation of...Ch. 5.5 - Prob. 12PCh. 5.6 - One of the following molecules (a)–(d) is...Ch. 5.6 - Prob. 14PCh. 5.6 - Assign R or S configuration to each chirality...Ch. 5.7 - Prob. 16PCh. 5.7 - Which of the following have a meso form? (Recall...Ch. 5.7 - Does the following structure represent a meso...Ch. 5.8 - Prob. 19PCh. 5.8 - Prob. 20PCh. 5.9 - Prob. 21PCh. 5.11 - Prob. 22PCh. 5.11 - Prob. 23PCh. 5.11 - The lactic acid that builds up in tired muscles is...Ch. 5.11 - The aconitase-catalyzed addition of water to...Ch. 5.SE - Which of the following structures are identical?...Ch. 5.SE - Prob. 27VCCh. 5.SE - Prob. 28VCCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 30VCCh. 5.SE - Prob. 31APCh. 5.SE - Which of the following compounds are chiral? Draw...Ch. 5.SE - Prob. 33APCh. 5.SE - Eight alcohols have the formula C5H12O. Draw them....Ch. 5.SE - Draw compounds that fit the following...Ch. 5.SE - Prob. 36APCh. 5.SE - Prob. 37APCh. 5.SE - Prob. 38APCh. 5.SE - What is the stereochemical configuration of the...Ch. 5.SE - Prob. 40APCh. 5.SE - Prob. 41APCh. 5.SE - Prob. 42APCh. 5.SE - Prob. 43APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 46APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configurations to the chirality...Ch. 5.SE - Assign R or S stereochemistry to the chirality...Ch. 5.SE - Prob. 50APCh. 5.SE - Draw examples of the following: (a) A meso...Ch. 5.SE - Prob. 52APCh. 5.SE - Prob. 53APCh. 5.SE - Prob. 54APCh. 5.SE - On reaction with hydrogen gas by a platinum...Ch. 5.SE - Prob. 56APCh. 5.SE - Prob. 57APCh. 5.SE - One of the steps in fat metabolism is the...Ch. 5.SE - The dehydration of citrate to yield cis-aconitate,...Ch. 5.SE - The first step in the metabolism of glycerol,...Ch. 5.SE - One of the steps in fatty-acid biosynthesis is the...Ch. 5.SE - Prob. 62APCh. 5.SE - Draw tetrahedral representations of the two...Ch. 5.SE - The naturally occurring form of the amino acid...Ch. 5.SE - Prob. 65APCh. 5.SE - Prob. 66APCh. 5.SE - Prob. 67APCh. 5.SE - Allenes are compounds with adjacent carbon-carbon...Ch. 5.SE - Prob. 69APCh. 5.SE - Prob. 70APCh. 5.SE - How many stereoisomers of...Ch. 5.SE - Draw both cis- and trans-1,4-dimethylcyclohexane...Ch. 5.SE - Draw both cis- and trans-1,3-dimethylcyclohexane...Ch. 5.SE - cis-1,2-Dimethylcyclohexane is optically inactive...Ch. 5.SE - Prob. 75APCh. 5.SE - Prob. 76APCh. 5.SE - Prob. 77AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Treatment of -D-glucose with methanol in the presence of an acid catalyst converts it into a mixture of two compounds called methyl glucosides (Section 25.3A). In these representations, the six-membered rings are drawn as planar hexagons. (a) Propose a mechanism for this conversion and account for the fact that only the OH on carbon 1 is transformed into an OCH3 group. (b) Draw the more stable chair conformation for each product. (c) Which of the two products has the chair conformation of greater stability? Explain.arrow_forwardThe dehydration of citrate to yield cis-aconitate, a step in the citric acid cycle, involves the pro-R “arm’’ of citrate rather than the pro-S arm. Which of the following two products is formed?arrow_forwardDraw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forward
- Give explanation for the following fact:- The enzyme alcohol dehydrogenase catalyzes the oxidation of CH3CH2OH to CH3CHO. But when racemic CH3CHDOH is similarly oxidized, an optically active enantiomer remains.arrow_forwardThe naturally occurring form of the amino acid cysteine (Problem 5-63) has the S configuration at its chirality center. On treatment with a mild oxidizing agent, two cysteines join to give cystine, a disulfide. Assuming that the chirality center is not affected by the reaction, is cystine optically active? Explain.arrow_forwardThe rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M-1s-1. In the presence of Co2+, the rate constant is 1.5 x 106 M-1s-1. What rate enhancement does the catalyst provide?arrow_forward
- Draw a mechanism for the conversion of glucose from its aldehyde form to its hemiacetal form and then to its methyl glycoside (acetal) form under acidic conditions. Show all bond breaking and bond forming steps; show all intermediates.arrow_forwardPGF2α is synthesized in cells using a cyclooxygenase enzyme that catalyzes a multistep radical pathway. Two steps in the pathway are depicted in the accompanying equations. (a) Draw in curved arrows to illustrate how C is converted to D in Step [1]. (b) Identify Y, the product of Step [2], using the curved arrows that are drawn on compound D.arrow_forwardThe chiral catalyst (R)-BINAP-Ru is used to hydrogenate alkenes to give alkanes . The products are produced with high enantiomeric excess. An example is the formation of (S)-naproxen, a pain reliever. Q.How can one enantiomer of naproxen be formed in such high yield?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License